Unconventional autophagy in health and disease
Research summary:
Autophagy is a degradative process that eliminates obsolete or potentially damaging cytoplasmic components and is critical for cellular homeostasis. Judging from the pathological phenotypes observed in mice lacking the expression of proteins that constitute the autophagic machinery (the so-called ATGs), autophagy has an important role in protection against a variety of pathologies, including cancer, inflammatory diseases or some neurodegenerative disorders. This protective capacity suggests that manipulation of autophagy could hold therapeutic value, a possibility that is being intensely studied by a variety of laboratories around the world. However, it is nowadays becoming clear that the different ATGs also have alternative functions not strictly related to the canonical autophagic pathway, either because their final purpose is not degradative, or because the relevant ATGs, even promoting catabolic outcomes, function in unconventional ways that are fundamentally different from autophagy. The contribution of these alternative activities to preventing the pathological phenotypes caused by ATG elimination is currently unclear, but their characterization is an important issue because it could determine the way ATG activity could be manipulated with therapeutic purposes.
With this overarching goal in mind, our lab focuses on ATG16L1 as a paradigm to discover and characterize new unconventional activities of the autophagic machinery. In its most canonical function, ATG16L1 is critical for proper formation of autophagosomes, double-membrane vesicles that sequester the cellular material that will eventually be degraded in the lysosome through the autophagic route. However, ATG16L1 includes a C-terminal domain that is irrelevant for this activity and seems to play unconventional roles in a wide variety of cellular processes. Projects currently being developed in the lab intend to discover and characterize in detail novel unconventional functions of ATG16L1 and other members of the general autophagic machinery, and explore how they interact with the conventional pathway. Current areas of interest in the lab include:
- Regulation of endocytosis, intracellular trafficking and signaling output of cytokine receptors
- Control of the stability of signaling mediators and the regulation of inflammatory responses
- Regulation of immunogenic cell death and its impact in cancer and response to chemotherapy
- Innate immunity against intracellular bacterial infections
To characterize these processes we use a wide variety of experimental systems include in vitro (cell lines) as well as in vivo (animal models) genetically engineered using the CRISPR/Cas9 system, proteomics techniques and bioinformatics approaches to identify novel mediators. We are currently trying to characterize in depth these mechanisms, discover novel non-canonical activators of ATG16L1 and explore how all these activities may be influenced by a coding polymorphic allele of ATG16L1 (T300A) whose presence in homozygosis increases the risk of suffering different pathologies, including Crohn’s disease, a serious intestinal inflammatory disease that is currently incurable. In this particular context, we intend to explore the molecular bases of complex, multifactorial diseases caused by the T300A allele, like Crohn’s disease. Exploring and establishing ways to manipulate atypical autophagy with therapeutic intentions is also a central long-term goal of our group that we intend to develop over the next few years.
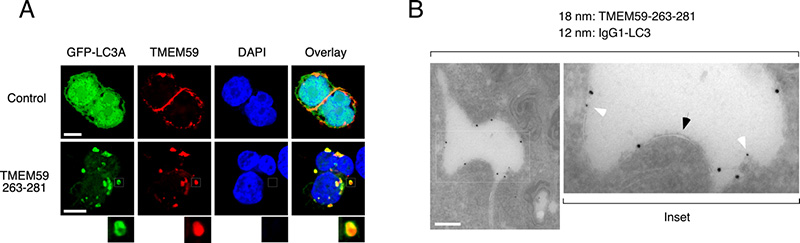
Figure 1. A. Confocal microscopy images showing the identification of a minimal active peptide in the transmembrane protein TMEM59 (residues 269-281) able to induce the unconventional autophagic labeling of the same endocytic vesicles where TMEM59 is expressed. B. Electron microscopy images showing that the LC3-labeled vesicles include just one membrane, and therefore are fundamentally different from canonical double-membrane autophagosomes.
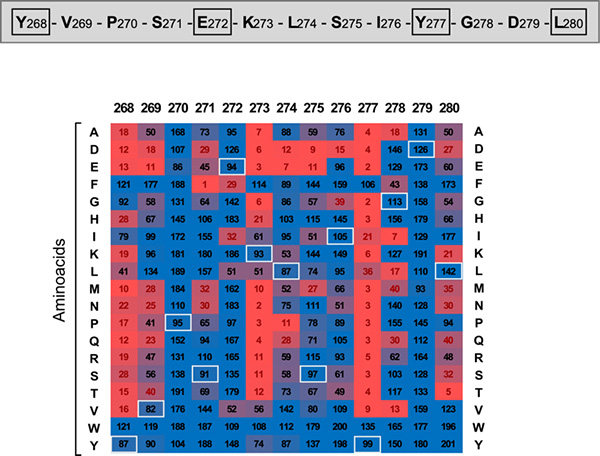
Figure 2. Mutagenic analysis based on peptide microarrays of the interaction between the 269-281 peptide and the WD40 domain of ATG16L1 in order to identify the critical residues and establish a consensus motif that mediates this interaction. The native peptide is shown on top. The heatmap shown below includes binding values expressed as the percentage of binding to the wild-type peptide that is retained by each mutant. Figures in black and red indicate permissive or prohibited substitutions, respectively. White squares highlight native residues for each position. All positions are colored following a continuous scale from low (red) to high (blue) binding activity.
LAB POSITIONS FOR TFG, TFM AND PhD THESIS: Two annual positions are available for candidates with good academic grades, highly motivated and interested in investigating the mechanisms that regulate unconventional autophagy and their role in health and disease. Interested candidates should contact the Principal Investigator (fxp@cbm.csic.es).

| Last name | Name | Laboratory | Ext.* | Professional category | |
|---|---|---|---|---|---|
| Bandera Linero | Julia | 411 | 4738 | julia.bandera(at)cbm.csic.es | Titulado Sup. Actividades Tecn. y Prof.GP1 |
| Oña Sánchez | Daniel | 411 | 4737 | donia(at)cbm.csic.es | M3 66,66% |
| Pimentel Muiños | Felipe X. | 411 | 4737 | fxp(at)cbm.csic.es | E. Investigadores Científicos de Organismos Públicos |
| Terraza Silvestre | Elena | 411 | 4738 | eterraza(at)cbm.csic.es | M3 66,66% |
| Vioque Hacha | Blanca | 411 | 4738 | Estudiante TFM |
Relevant publications:
- Serramito-Gómez, I; Boada-Romero, E; Villamuera, R; Fernández-Cabrera, A; Cedillo, JL; Martín-Regalado, A; Carding, S; Mayer, U; Powell, PP; Wileman, T; García-Higuera, I; Pimentel-Muiños, FX. (2020). Regulation of cytokine signaling through direct interaction between cytokine receptors and the ATG16L1 WD40 domain. Nat. Commun., 11:5919.
- Serramito-Gómez, S; Boada-Romero, E; Slowicka, K; Vereecke, L; Van Loo, G; Pimentel-Muiños, FX. (2019). The anti-inflammatory protein TNFAIP3/A20 binds the WD40 domain of ATG16L1 to control the autophagic response, NF-kB activation and intestinal homeostasis. Autophagy, 15:1657.
- Slowicka, K; Serramito-Gómez, S; Boada-Romero, E; Martens, A; Sze, M; Petta, I; Vikkula, HK; De Rycke, R; Parthoens, E; Lippens, S; Savvides, SN; Wullaert, A; Vereecke, L; Pimentel-Muiños, FX(*); van Loo, G(*). (2019). Physical and functional interaction between A20 and ATG16L1-WD40 domain in the control of intestinal homeostasis. Nat. Commun., 10:1834. (*) Co-corresponding senior authors
- Gerlach, JP; Jordens, I; Tauriello, DVF; van’t Land-Kuper, I; Bugter, JM; Noordstra, I; van der Kooij, J; Low, TY; Pimentel-Muiños, FX; Xanthakis, D; Fenderico, N; Rabouille, C; Heck, AJR; Egan, DA; Maurice, MM. (2018). TMEM59 potentiates Wnt signaling by promoting signalosome formation. PNAS., 115:E3996.
- Serramito-Gómez, I; Boada-Romero, E; Pimentel-Muiños, FX (2016). Unconventional autophagy mediated by the WD40 domain of ATG16L1 is derailed by the T300A Crohn disease risk polymorphism. Autophagy, 12:2254-2255.
- Boada-Romero, E; Serramito-Gómez, I; Sacristán, MP; Boone, DL; Xavier, RJ; Pimentel-Muiños, FX (2016). The T300A Crohn’s disease risk polymorphism impairs function of the WD40 domain of ATG16L1. Nat. Commun, 7:11821.
- Pimentel-Muiños, FX; Boada-Romero, E. (2014). Selective autophagy against membranous compartments: Canonical and unconventional purposes and mechanisms. Autophagy, 10:397-407.
- Boada-Romero, E; Letek, M; Fleischer, A; Pallauf, K; Ramón-Barros, C; Pimentel-Muiños, FX. (2013). TMEM59 defines a novel ATG16L1-binding motif that promotes local activation of LC3. EMBO J., 32:566-582.
- Rodríguez, DA; Zamorano, S; Lisbona, F; Rojas-Rivera, D; Urra, H; Cubillos-Ruiz, JR; Armisen, R; Henriquez, DR; Cheng, EH; Letek, M; Vaisar, T; Irrazabal, T; Gonzalez-Billault, C; Letai, A; Pimentel-Muiños, FX; Kroemer, G; Hetz, C. (2012). BH3-only proteins are part of a regulatory network that controls the sustained signaling of the unfolded protein response sensor IRE1a. EMBO J., 31:2322-2335.
- Klee, M; Pallauf, K; Alcalá, S; Fleischer, A; Pimentel-Muiños, FX. (2009). Mitochondrial apoptosis induced by BH3-only molecules in the exclusive presence of endoplasmic reticular Bak. EMBO J., 28:1757-1768




















