Mitochondrial biology in immune modulation
Research summary:
Mitochondria are known as the powerhouses of the cell, and yet their functions are much more complex. In addition to energy conversion, they are also involved in heat production, calcium signaling and storage, signaling through and detoxification of reactive oxygen species (ROS), synthesis of heme and other molecules, and regulation of cell death. Emerging functions of the mitochondria in disease include their role as damage-associated molecular patterns (DAMPs), which are important for innate and adaptive immune activation. In this context, release of mitochondrial components of bacterial origin, such as mitochondrial DNA, may activate several pathways that lead to the secretion of pro-inflammatory cytokines (Fig. 1), a phenomenon called sterile inflammation. Among them, the most studied pathway is the NLRP3 inflammasome, a cytosolic multiprotein complex that senses the presence oxidized mitochondrial DNA (mtDNA) in the cytosol (and thus acts as a sensor for mitochondrial dysfunction) and in turn activates caspase-1, an enzyme that cleaves other proteins, including the precursors of the inflammatory cytokines interleukin 1β and interleukin 18, as well as the pyroptosis inducer Gasdermin D, into active mature peptides. The NLRP3 inflammasome thus plays a central role in immunity as an inflammatory response initiator and is associated with a broad range of degenerative diseases associated with aging, including Alzheimer's, asthma, gout, ischemia/reperfusion, hypertension, diabetes or psoriasis.
Mitochondria are also being recognized as important nutrient-sensing organelles which functionally adapt in a nutrient-dependent manner. Nutrient restriction leads to activation of several pathways and to higher levels of intracellular nicotinamide adenine dinucleotide (NAD+), which activates sirtuin proteins, a group of enzymes possessing deacetylase activity that require NAD+ as a cosubstrate to function, and thus are considered metabolic and energy sensors.
Our research has shown that nutrient restriction blunted the activation of the inflammasome in macrophages, and that this effect depended partially on the activation of the NAD+-dependent mitochondrial deacetylase enzyme Sirtuin 3 (SIRT3). The mechanism of action of SIRT3 is very intriguing: by modulating the acetylation status and activity of mitochondrial superoxide dismutase (SOD2), and thus mitochondrial ROS levels, it finely controls the extrusion of oxidized mtDNA into the cytosol, where it acts as an NLRP3 agonist (Fig. 2). In addition, we have found that nicotinamide riboside (NR), an intermediate precursor of NAD+ synthesis in the salvage pathway, functions as a fasting mimetic and blunts monocyte/macrophage IL-1β production and reduces T helper 1 (Th1) and 17 (Th17) cell activation. Interestingly, a mouse model of psoriasis, a chronic inflammatory skin disease linked to hyperactivation of Th17 cells, displayed downregulation of the SIRT3 gene and decreased mitochondrial SOD2 activity, and thus can be considered a functional SIRT3 knockdown. The downregulation of this gene might be involved in the hyperinflammatory phenotype observed in some of its tissues. It is also known that NLRP3 inflammasome activation and IL-1β signaling are associated with psoriasis progression. Given our findings of the role of SIRT3 in immune-modulation, a question arising is whether NAD+ precursors could mimic caloric restriction effects and ameliorate an inflammatory disease. Psoriasis, a prototypic Th17 disease, appears to be an appealing candidate disease to test this hypothesis.
The main lines of research of our group are the following:
- Expand our studies into the fundamental role of mtDNA in innate inflammatory pathways regulated by the mitochondrial protein SIRT3, the main deacetylase in the mitochondria.
- Evaluate whether NAD+ precursors blunt inflammation in a psoriatic mouse model via augmentation of mitochondrial function, fidelity and quality control programs.
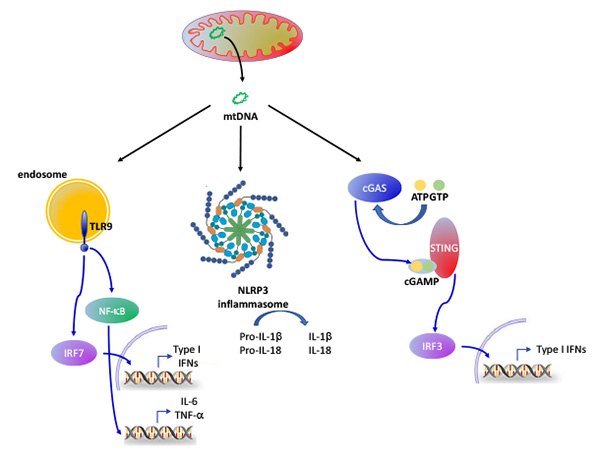
Fig.1: Release of mtDNA during mitochondrial dysfunction triggers at least three distinct pathways linked to inflammation: the endosomal TLR9 pathway, the NLRP3 inflammasome and the cGAS-STING pathway.
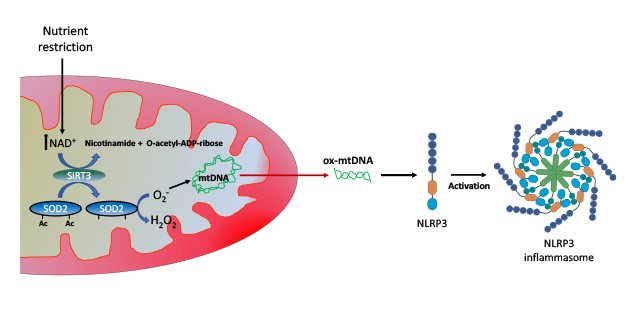
Fig. 2: Mechanism of action of SIRT3 in immune modulation: nutrient restriction increases the levels of NAD+, which activates the mitochondrial deacetylase SIRT3, that in turn deacetylates and activates SOD2. SOD2 decreases mitochondrial levels of superoxide, and therefore protects mitochondrial DNA from oxidation and release to the cytosol, where it might act as an agonist of NLRP3.

| Last name | Name | Laboratory | Ext.* | Professional category | |
|---|---|---|---|---|---|
| Cuesta Valero | Yara | 321 | 4651 | ycuesta(at)cbm.csic.es | M3 Predoc.formación |
| Meroño Ortega | Carolina | 321 | 4651 | cmeronio(at)cbm.csic.es | Titulado Sup.de Actividades Técn. y Profes. GP1 |
| Rincón Álvarez | Rubén | 321 | 4651 | Estudiante TFG | |
| Traba Domínguez | Javier | 321 | 4622 | jtraba(at)cbm.csic.es | Investigador Doctor |
Relevant publications:
- Traba J., Waldmann T.A., Anton O.M. (2020) Analysis of human Natural Killer Cell metabolism. J Vis Exp (160), doi: 10.3791/61466.
- Anton O.M., Peterson M.E., Hollander M.J., Dorward D.W., Arora G., Traba J., Rajagopalan S., Snapp E., Garcia K.C., Waldmann T.A., Long E.O. (2020) Trans-endocytosis of intact IL-15Rα-IL-15 complex from presenting cells into NK cells favors signaling for proliferation. Proc Natl Acad Sci USA 117, 522-531.
- Akkaya M.1, Traba J.1, Roesler A.S., Miozzo P., Akkaya B., Theall B.P., Sohn H., Pena M., Smelkinson M., Kabat J., Dahlstrom E., Dorward D., Sack M.N., Pierce S.K. (2018) Second signals rescue B cells from activation-induced mitochondrial dysfunction and death. Nat Immunology 19, 871-884. (1: Equal contribution).
- Traba J., Geiger S.S., Kwarteng-Siaw M., Han K., Ra O.H., Siegel R.M, Gius D., Sack M.N. (2017) Prolonged fasting suppresses mitochondrial NLRP3 inflammasome assembly and execution via SIRT3 mediated activation of SOD2. J Biol Chem 292, 12153-12164.
- Traba J., Sack M.N. (2017) The role of caloric load and mitochondrial homeostasis in the regulation of the NLRP3 inflammasome. Cell Mol Life Sci 74, 1777-1791.
- Traba J., Kwarteng-Siaw M., Okoli T.C, Li J., Huffstutler R.D., Bray A., Waclawiw M.A., Han K., Pelletier M., Sauve A.A., Siegel R.M, Sack M.N. (2015) Fasting and refeeding differentially regulate NLRP3 inflammasome activation in human subjects. J Clin Invest 125, 4592-4600.
- Rueda C.1, Traba J.1, Amigo I.1, Llorente-Folch I., Gonzalez-Sanchez P., Pardo B., Esteban J.A., del Arco A., and Satrustegui J. (2015) Mitochondrial ATP-Mg/Pi carrier SCaMC-3/Slc25a23 counteracts PARP-1-dependent fall in mitochondrial ATP caused by excitotoxic insults in neurons. J Neurosci 35, 3566-3581. (1: Equal contribution).
- Webster B.R., Scott I., Traba J., Han K., and Sack M.N. (2014) Caloric Restriction, Acetylation and the Regulation of Autophagy and Mitophagy. Biochim Biophys Acta 1841, 525-534.
- Traba J., del Arco A., Duchen M.R., Szabadkai G. and Satrústegui J. (2012) SCaMC-1 promotes cancer cell survival by desensitizing mitochondrial permeability transition via ATP/ADP-mediated matrix Ca2+ buffering. Cell Death Differ 19, 650-660.
- Traba J.1, Satrústegui J. and del Arco A. (2011) Adenine nucleotide transporters in organelles: novel genes and functions. Cell Mol Life Sci 68, 1183–1206. (1: Corresponding author).
Funding:
- Ministerio de Ciencia e Innovación (MICINN) :
- Ramón y Cajal 2018 (RYC2018-026050-I)
- Programa Estatal de I+D+i Orientada a los Retos de la Sociedad (PID2019-105665RA-I00)




















