Cell polarity
Research summary:
Cell polarity is fundamental to the functioning of most types of cell. Our group's traditional goal is to advance our knowledge of the cell polarization process through the functional characterization of protein machinery involved in the generation and maintenance of cell polarization. Hepatocytes, T lymphocytes and epithelial cells are our preferred model cell systems. More recently, our group is also interested in studying the mechanism of biogenesis of the primary cilia by polarized epithelial cells and analyzing the role of the formin INF2, a protein whose mutation causes kidney disease with or without associated peripheral neuropathy.
The primary cilium is a single appendage that projects from the surface of most mammalian cells. One of the main features of primary cilia is that, unlike the motile cilia of multiciliary cells or of cells that use cilia or flagella for movement, they are non-motile. Important signaling pathways involved in cell proliferation, differentiation, survival and migration, such as Hedgehog, Wnt, Notch, signaling, are orchestrated in the primary cilium. Dysfunction of the cilium is associated with a long list of human developmental and degenerative disorders, collectively referred to as ciliopathies, that affect nearly every major body organ. The midbody remnant is an electrodense structure derived from the intercellular bridge that connects tthe two daughter cells during cytokinesis. Our laboratory has recently established that in polarized epithelial MDCK cells, the midbody remnant moves to the center of the apical surface to meet the centrosome and licenses it to form a primary cilium. We have begun to investigate: 1) what the remnant does to enable primary cilium biogenesis, and 2) how the inheritance of the remnant is regulated.
Formins are an extended family of proteins involved in the formation of linear polymers of actin. In addition to this role, formins bind to microtubules and regulate their stability. Mutations in the formin INF2 are responsible for two specific types of human degenerative hereditary diseases that affect the kidney (focal segmental glomerulosclerosis) and the peripheral nerves (Charcot-Marie-Tooth disease). We have found that INF2 controls the acetylation and the detyrosination of the microtubules, two posttranslational modifications that are frequently altered in degenerative diseases and cancer. We are now interested in investigating the role of these modifications in INF2-related disease.
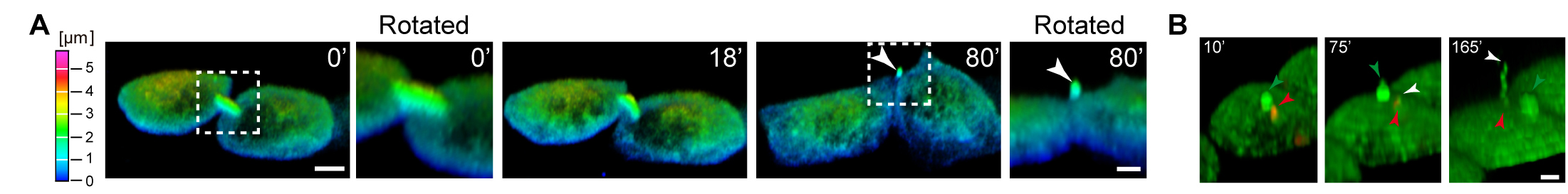
Figure 1. The midbody remnant enables the centrosome for primary cilium formation. (A) The images correspond to3D reconstructions of cells stably expressing cherry-tubulin that were filmed during cell division.The images were pseudocolored based on height using the color scale on the left to highlight that the intracellular bridge forms at the top of the cells and that the post-mitotic midbody remnant localizes after abcission at a peripheral position at the apical surface. The arrowhead points to the post-mitotic midbody. An enlargement of the boxed region at 0 min and 80 min is also shown. (B) Three-dimensional reconstruction of images obtained by videomicroscopic analysis of cells expressing GFP-tubulin (midbody remnant and primary cilium) and dsRed-centrin (centrosome) during the movement of the midbody to encounter the centrosome for primary cilium formation. The green and red arrowheads point to the midbody remnant and the centrosome, respectively.
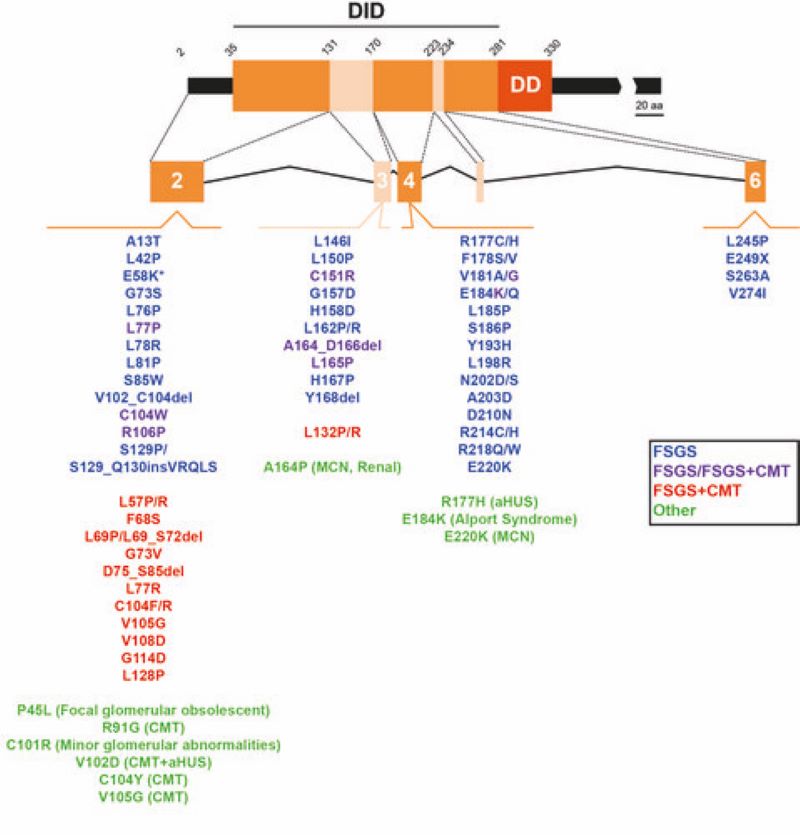
Figure 2. Pathogenic mutations of INF2. The exons encoding different segments of the human diaphanous inhibitory domain (DID) of INF2 are indicated. The INF2 mutations associated with isolated FSGS (blue), FSGS in some patients or FSGS + CMT in others (purple), FSGS + CMT (red), and other diseases (green) are listed.

| Last name | Name | Laboratory | Ext.* | Professional category | |
|---|---|---|---|---|---|
| Alonso Lebrero | Miguel Ángel | 322 | 4614 | maalonso(at)cbm.csic.es | E. Profesores de Investigación de Organismos Públicos de Investigación |
| Fernández Martín | Laura | 322 | 4644/45 | lfernandez(at)cbm.csic.es | M1 |
| Labat de Hoz | Leticia | 322 | 4645 | llabat(at)cbm.csic.es | Tit.Sup.Activ.Técn.y Profes. GP1 |
Relevant publications:
-
Andres-Delgado, L., Anton, O. M., Bartolini, F., Ruíz-Saénz, A, Correas, I., Gundersen, G. G., and Alonso, M. A. (2012) INF2 promotes the formation of detyrosinated microtubules necessary for reorientation of the centrosome to the immunological synapse of T cells. J. Cell Biol. 198, 1025-1037.
-
Reales, E., Bernabé-Rubio, M., Casares-Arias, J., Rentero, C., Fernández-Barrera, J., Rangel, L., Correas, I., Enrich, C., Andrés, G. and Alonso, M.A. (2015) The MAL protein is crucial for proper membrane condensation at the ciliary base, which is required for primary cilium elongation. J. Cell Sci. 128, 2261-2270.
-
Bernabé-Rubio, M., Andrés, G., Casares-Arias, J., Fernández, J.J., Fernández-Barrera, J., Rangel, L., Reglero-Real, N., Gershlick, D.C., Fernández, J.J., Millán, J., Correas, I., Miguez, D.G., and Alonso, M.A. (2016) Novel role for the midbody in primary ciliogenesis by polarized epithelial cells. J. Cell Biol. 214, 259-273.
-
Fernández-Barrera, J., Bernabé-Rubio, M., Casares-Arias, J., Rangel, L., Fernández-Martín, L., Correas, I., and Alonso, M.A. (2018) The actin-MRTF-SRF transcriptional circuit controls tubulin acetylation via a-TAT1 gene expression. J. Cell Biol. 217, 929-944.
-
Fernández-Barrera, J., and Alonso, M.A. (2018) Coordination of microtubule acetylation and the actin cytoskeleton by formins. Cell. Mol. Life Sci. 75, 3181-3191. doi:10.1007/s00018-018-2855-3
-
Labat-de-Hoz, L., and Alonso, M. A. (2020) The formin INF2 in disease: progress from 10 years of research. Cell. Mol. Life Sci. 77, 4581-4600. doi: 10.1007/s00018-020-03550-7.
-
Casares-Arias, J., González, M.U., San Paulo, A., Ventimiglia, L.N., Sadler, J.B.A., Miguez, D.G., Labat-de-Hoz, L., Rubio-Ramos, A., Rangel, L., Bernabé-Rubio, M., Fernández-Barrera, J., Correas, I., Martín-Serrano, J., and Alonso, M.A. (2020) Midbody remnant inheritance is regulated by the ESCRT subunit CHMP4C. iScience 23: 101244. doi: 10.1016/j.isci.2020.101244
-
Bernabé-Rubio, M., Bosch-Fortea, M., García, E., Bernardino de la Serna, J., and Alonso, M.A. (2021) Adaptive lipid immiscibility and membrane remodeling are active functional determinants of primary ciliogenesis. Small Methods. 5:2000711. doi: 10.1002/smtd.202000711
-
Labat-de-Hoz, L., Rubio-Ramos, A., Casares-Arias, J., Bernabé-Rubio, M., Correas, I., and Alonso, M.A. (2021) A model for primary cilium biogenesis by polarized epithelial cells: Role of the midbody remnant and associated specialized membranes. Front. Cell Dev. Biol. 8:622918. doi: 10.3389/fcell.2020.622918
More information at https://orcid.org/0000-0002-7001-8826




















