Group of intestinal morphogenesis and homeostasis
Research summary:
Our main scientific interest is the understanding of intestinal morphogenesis and cellular polarity during morphogenesis, homeostasis, and regeneration, as well as their implications in human diseases, such as intestinal bowel diseases (IBD), obesity, diabetes, and cancer. Our research is based in the organotypic culture model of three-dimensional epithelial cells growing in micropatterns “organ-on-a-chip”, which are becoming one of the best in vitro models systems for the investigation of epithelial morphogenesis. Moreover, with this system, we are obtaining essential information about the molecular mechanisms that regulate epithelial morphogenesis. However, this model cannot reconstitute the complexity of the architecture given in vivo, which includes different cell types, dynamic remodeling, and tissue homeostasis. For this reason, the use of in vivo systems should serve to validate and better characterize the phenotypes observed in vitro. We used the zebrafish and mouse intestine as models systems to elucidate epithelial morphogenesis and intestinal homeostasis.
We are focused on the analysis of genes that regulate epithelial polarity during morphogenesis, B cell response and intestinal homeostasis, and particularly those controlling the following processes: Signaling, membrane trafficking, mechanical forces, and metabolic remodeling. Therefore, our lab is focused on:
- Characterization of signaling pathways for epithelial morphogenesis, differantiation and patterning
- Analysis of cellular mechanics controlling lumen formation in epithelial cells
- Study of the metabolic reprogramming that takes place along intestinal development and homeostasis.
- Characterization of the metabolic crosstalk among microbiota, epithelial cells, and immune cells controlling the intestinal homeostasis.
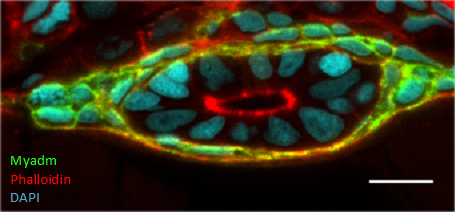
Figure 1. Epithelial morphogenesis in the zebrafish intestine. A gut cross-section of a two-day post-fertilization larva is shown after co-staining to visualize F-actin (red), myadm-GFP (green, myoepithelial marker), and nuclei (light blue).
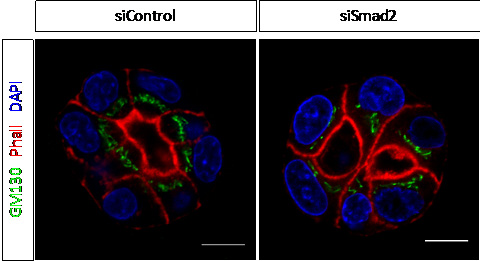
Figure 2. Epithelial morphogenesis in the 3D organotypic models. Confocal sections of a 3-day organoid KD for Smad3 or control are shown after staining to visualize F-actin (red), GM130 (green) and nuclei (blue).
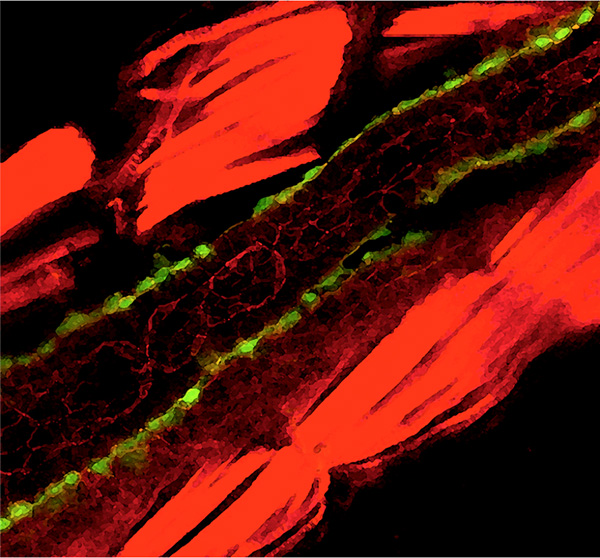
Figure 3. An artistical version of epithelial morphogenesis in the zebrafish intestine. A transverse gut section of a 3-day post-fertilization larva is shown after co-staining to visualize F-actin (epithelial cells, red) and Patch-GFP (myoepithelial cells, green).

| Last name | Name | Laboratory | Ext.* | Professional category | |
|---|---|---|---|---|---|
| Akintche | Laura | 120 | 4721 | M3 Predoc.formación | |
| Alfonso Pérez | Tatiana | 120 | 4721 | talfonso(at)cbm.csic.es | Investigador |
| Alonso Larre | Diego | 120 | 4721 | diego.alonso(at)cbm.csic.es | Ayudante Investigación |
| Baena López | Luis Alberto | 4401 | labaena(at)cbm.csic.es | E. Investigadores Científicos de Organismos Públicos | |
| Baonza Martínez | Gabriel | 120 | 4615 | gbaonza(at)cbm.csic.es | Titulado Sup. Actividades Tecn. y Prof.GP1 |
| González Martínez | Tamara | 120 | 4721 | tgonzalez(at)cbm.csic.es | Técnico Sup. Actividades Tec. y Profes.GP3 |
| Martín Belmonte | Fernando | 120 | 4615 | fmartin(at)cbm.csic.es | E. Profesores de Investigación de Organismos Públicos de Investigación |
Relevant publications:
- Hachimi M, Grabowski C, Campanario S, Rodriguez-Fraticelli A, Gomez-Lopez S, and Martin-Belmonte F. A network of actomyosin regulators controls apical maturation in epithelia. Current Biol (in 2nd revision).
- Bosch-Fortea M, Rodriguez-Fraticelli A, Herranz G, Hachimi M, Ladoux B, Martin-Belmonte F. (2019) Micropattern-Based Platform as a Physiologically Relevant High-Throughput Model to Predict Nephrotoxicity of New Drugs. Biomaterials 18:119339.
- Bernascone I, Gonzalez T, Delgado M, Carabaña C, Hachimi M, Bosch-Fortea M, Santamaría S, Martin R, Tarnick J, García-Sanz JA, and Martin-Belmonte, F. (2019) Sfrp3 modulates stromal–epithelial crosstalk during mammary gland development by regulating Wnt levels. Nature Comm 10(1):2481.
- Bosch-Fortea M, and Martin-Belmonte F (2018) Mechanosensitive adhesion complexes in epithelial architecture and cancer onset. Current Opinion in Cell Biology 50:42–49
- Bernascone, I; Hachimi, M and Martin-Belmonte, F (2017) Signaling networks in epithelial tube formation and regeneration. Cold Spring Harbor Perspectives Biol. pii: a027946.
- Fütterer A, de Celis J, Navajas R, Almonacid L, Gutiérrez J, Talavera-Gutiérrez A, Pacios-Bras C, Bernascone I, Martin-Belmonte F, Martinéz-A C. (2017) DIDO as a Switchboard that Regulates Self-Renewal and Differentiation in Embryonic Stem Cells. Stem Cell Reports. 8(4):1062-1075.
- Tsui C, Maldonado P, Montaner B, Borroto A, Alarcon B, Bruckbauer A, Martinez-Martin N, Batista FD. (2018) Dynamic reorganisation of intermediate filaments coordinates early B-cell activation. Life Sci Alliance. 1(5):e201800060.
- Martínez-Riaño A, Bovolenta ER, Mendoza P, Oeste CL, Martín-Bermejo MJ, Bovolenta P, Turner M, Martínez-Martín N, Alarcón B. (2018) Antigen phagocytosis by B cells is required for a potent humoral response. EMBO Rep. 19(9).
- Tsui C*, Martinez-Martin N*, Gaya M, Maldonado P, Llorian M, Legrave NM, Rossi M, MacRae JI, Cameron AJ, Parker PJ, Leitges M, Bruckbauer A, Batista FD. (2018) Protein Kinase C-β Dictates B Cell Fate by Regulating Mitochondrial Remodeling, Metabolic Reprogramming, and Heme Biosynthesis. Immunity; 48(6):1144-1159.e5. (*equally contributed).
- Mendoza P*, Martínez-Martín N*, Bovolenta ER, Reyes-Garau D, Hernansanz-Agustín P, Delgado P, Diaz-Muñoz MD, Oeste CL, Fernández-Pisonero I, Castellano E, Martínez-Ruiz A, Alonso-Lopez D, Santos E, Bustelo XR, Kurosaki T, Alarcón B. R-(2018) Ras2 is required for germinal center formation to aid B cells during energetically demanding processes. Sci Signal. 11(532). (*equally contributed).
- Martinez-Martin N, Maldonado P, Gasparrini F, Frederico B, Aggarwal S, Gaya M, Tsui C, Burbage M, Keppler SJ, Montaner B, Jefferies HB, Nair U, Zhao YG, Domart MC, Collinson L, Bruckbauer A, Tooze SA, Batista FD (2017) A switch from canonical to noncanonical autophagy shapes B cell responses. Science. 355(6325):641-647.
Doctoral theses:
- Title: Adress the role of non-canonical autophagy in epithelial polarity and morphogenesis. PhD Student: Sergio Gómez Lopez. University: Universidad Autónoma de Madrid. Faculty: CBMSO. Date: 18/12/2019.
- Title: Exploring the roles of major signaling pathways in epithelial morphogenesis. PhD student: Maria D. Barea. University: Universidad Autónoma de Madrid. Faculty: CBMSO. Date: 14/10/ 2019.
- Title: The role of SMTL2 in zebrafish epithelial morphogenesis. PhD student: Mariam Hachimi. University: Universidad Autónoma de Madrid. Faculty: CBMSO. Date: May/ 2018.
- Title: Development of a 3D micropattern-based tube model system to study epithelial morphogenesis, tube mechanics and nephrotoxicity. PhD Student: Minerva Bosch Fortea. University: Universidad Autónoma de Madrid. Faculty: CBMSO. Date: April/2018.
Other relevant scientific activities and recognitions:
- 2018 Organizer of the meeting on Cell compartment and transport. CBMSO, Madrid
- 2017 Organizer of the EMBO meeting on Cell polarity and membrane dynamics. Sant Feliu de Gixols, Girona
- 2015 Present -Associated editor of Nephron (Journal).
- 2015 Present- Scientific vice deputy of Spanish agency of research (Agencia Estatal Investigación) (AEI-MINECO-Area BFS)
- Master professor –Coordination of sorting, routing and distribution of proteins in polarized cells, Universidad Autonoma de Madrid/ Madrid/ Spain.
- Master professor –Molecular Biology of the Cell course at Pasteur, Institute Pasteur
- Master professor –Cytoskeleton course at Institute Curie/ Paris/ France




















