Mechanisms of Tumorigenesis in Drosophila
The main focus of our research is the study of tumorigenesis, a phenomenon of great scientific and societal relevance. Because of its sophisticated genetic technology and other experimental advantages, we are using the imaginal discs of the fruitfly Drosophila melanogaster as a model system to investigate molecular and genetic factors behind the appearance and development of tumors. We are focusing on three research lines.
1)The tumorigenic potential of cells that cannot execute the apoptosis program.
This stands from previous published work by our team about the behavior of cells in which the apoptosis machinery is suppressed; these Apoptosis-Deficient (AD) cells survive after stress or drug treatments that lead to death in normal cells. We have developed a model of tumorigenesis based on the properties of those cells: after a transient stress, a fraction of the AD population activates ectopically the pro-proliferation and pro-motility functions of the Jun N-Terminal Kinase (JNK) pathway, what results in the generation of invasive tumor overgrowths. The development of those tumors is strictly mediated by JNK activity. It is worth mentioning that inappropriate activity of the JNK pathway is also associated with human tumors.
Given that JNK appears to be the key factor in the oncogenic effect, we are studying the mechanisms by which AD cells expressing JNK (figure 1) drive tumor formation. We have found that they display all the characteristic features of senescent cells, including the Senescence Associate Secretory Phenotype (SASP). In particular, they secrete the signaling molecules Dpp, Wg and Upd, the ligands that trigger the Dpp, Wg and JAK/STAT pathways respectively. Experiments in which the activity of those pathways is suppressed indicate that their functions are responsible for the tumorous overgrowths, although the specific effect of each pathway and their interactions are not yet known and are under study in the lab. We also have preliminary evidence that not all senescent cells acquire the SASPhenotype; those with SASP appear to localize to the center of the disc, while those in the notum or in the peripheral regions do not express the SASPhenotype. The finding that senescent cells come in two classes, secretory (with SASP) and non-secretory (without SASP), is novel in the field. It correlates with the observation that the tumorous overgrowths appear uniquely in the center of the disc where the secretory senescent cells localize. The mechanisms behind this positional effect are under intense scrutiny in the laboratory.
A related issue is how JNK activity is able to perform all those activities. Some years ago, we performed a RNAseq screen seeking genes specifically activated by JNK and necessary for the overgrowths. Several of the genes identified in the screen are currently being tested for their implications in the signaling emanating from the senescent cells and the overproliferation they produce.
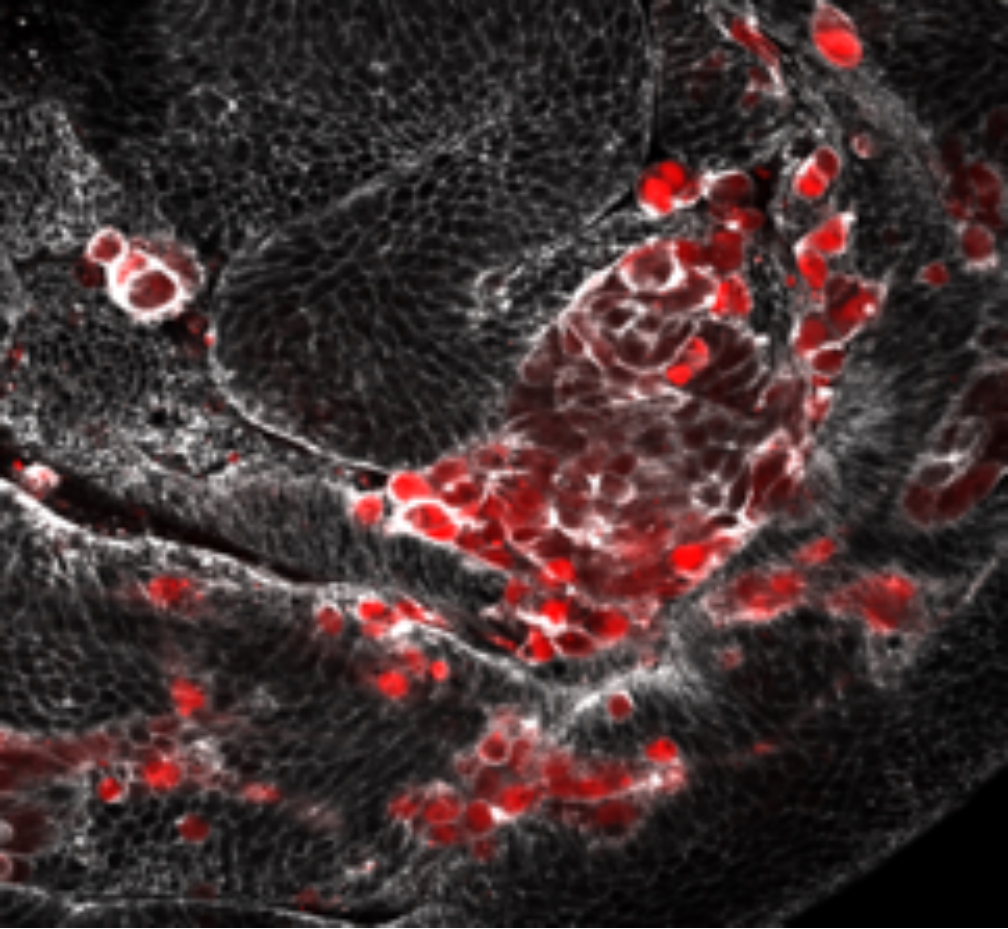
Figure 1. Portion of an imaginal disc of Drosophila containing senescent cells (in red). The outline of all cells is delineated by phalloidin. Note that senescent cells are bigger than non-senescent cells
2) Chromatin modifications of senescent cells and reversibility of senescence
Senescence can be induced by a range of cellular stresses (DNA damage, oxidative stress, oncogene activation) that cause profound, usually stable, alterations in cell function and structure. One marker of the transformation is a chromatin structural change, referred to as senescence-associated heterochromatic foci (SAHFs). Upon the induction of senescence in mammalian cells, SAHFs become visible as foci enriched in heterochromatin markers (as H3K9me3) and diminished in euchromatin ones. We are in the first steps of a description of the process of chromatin reshuffling during the transformation of normal into senescent cells, which has not been carried out in Drosophila. We have already found that, as described in vertebrate culture cells, Drosophila wing disc cells increase their nucleoli size when becoming senescent. The levels of H3K9me3 are also higher in these cells. Since JNK is an inducer of senescence, we are currently studying how JNK activity is resolved into those chromatin alterations. Some of the JNK targets identified in the RNA screen mentioned above have been associated with chromatin modifications (such as Caf1-105, involved in heterochromatin formation and maintenance). We are analysing the possible implication of some of those genes in the chromatin modifications associated with senescence.
In addition, we have found an unexpected role of F-element retrotransposons (that belong to the LINE family) in the maintenance of the senescent state; when they are expressed at high levels senescent cells appear to revert to normal state (Figure 2). This is a very significant observation, first because it reveals that these elements – known to be involved in chromatin remodeling during early embryogenesis in the mouse and Drosophila - have a role in the senescent state, and second because it is the first indication that the senescent status can be reversed. The molecular details of this phenomenon are being currently pursued.
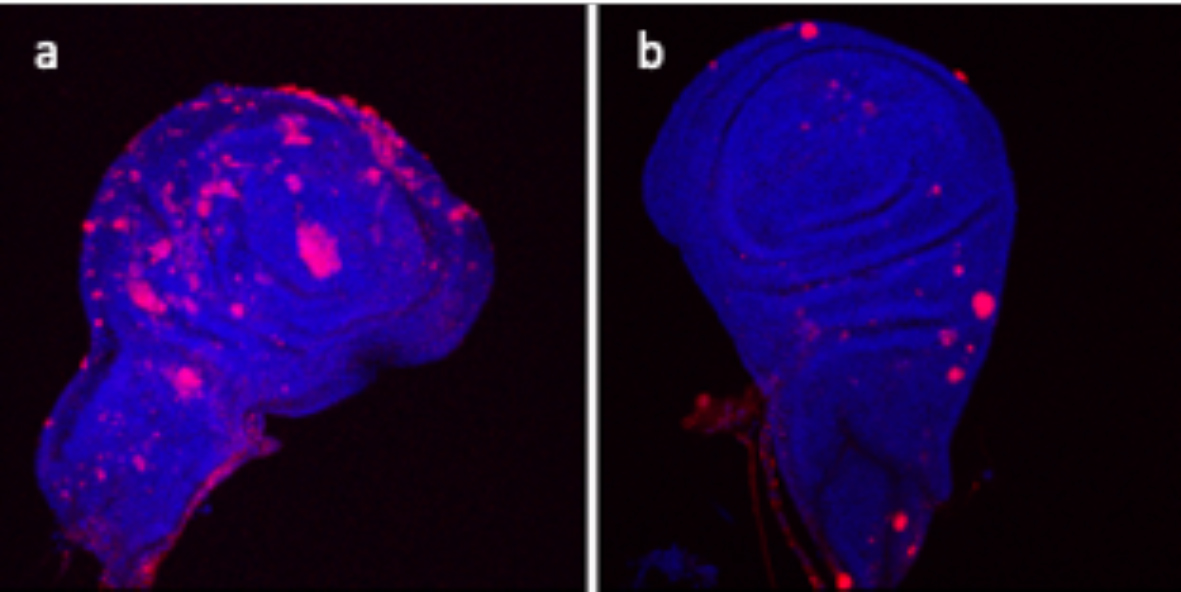
Figure 2. a) Wing imaginal disc of Drosophila that has been irradiated and has developed senescent cells (in red). b) Wing imaginal disc treated as in a) but with overexpression of the retrotransponson F-element. Note that the number of senescent cells is very much reduced.
3) Interactions among cell populations with different growth rates
In our analysis of the tumorous overgrowths generated by oncogenic mutations at polyhomeotic (ph) and erupted (the Drosophila homologue of the mammalian tumor susceptibility gene 101), we have made a novel observation that may have implications in the understanding of how tumor cells colonize adjacent normal tissues: in experiments in which one compartment of the wing disc is of normal genotype and the overgrowing adjacent one is mutant for the oncogenic gene ph or erupted the normal one becomes smaller than its normal size (figure 3).
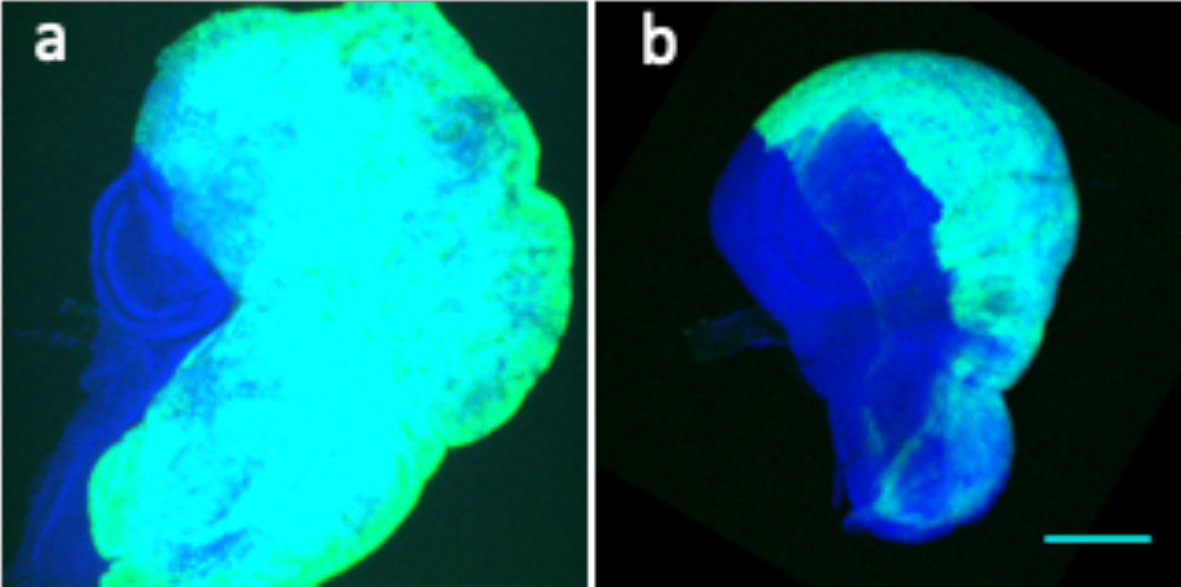
Figure 3. a) Wing disc of genotype hh-Gal4> UAS-phi UAS-GFP. The posterior compartment (green) is overgrown while the anterior compartment (blue) is of normal genotype. Notice that the size of the anterior compartment is clearly smaller than normal. Compare with its size that in the disc in b) in which the posterior compartment in not overgrown. The two photographs are taken at the same magnification
We have observed that this phenomenon also occurs among regions within the same compartment. It appears that overgrowing cell populations inhibit growth of neighbor tissues. Tentatively we refer to those populations as “winner” (the overgrowing one) and “loser” (the undergrowing one). We have already ruled out the hypothesis of an increase of apoptosis in the loser population, but found that the loser populations have a mitotic index lower than normal, which explains their reduced growth. Since the overall growth of the disc depends on signaling growth factors like Dpp or Wg, we are examining the possibility that the cells of the winner populations may sequester the Dpp or Wg signals, thus preventing their accessibility to the loser population.
We are trying to identify the genetic factors associated with the reduction of the mitotic index, examining the expression levels of genes regulating cell division, either those promoting division (cdk, other cyclin dependent kinases) or inhibiting it (dacapo, frs). There is also the possibility of up regulation of the Hippo pathway, a well characterized cell proliferation inhibitor, which can be tested using the numerous probes (expanded, warts, Yki) available to monitor its activity.

| Last name | Name | Laboratory | Ext.* | Professional category | |
|---|---|---|---|---|---|
| Calleja Requena | Manuel | 422.1 | 4697/4683 | mcalleja(at)cbm.csic.es | E.Científicos Titulares de Organismos Públicos de Investigación |
| Cantarero Mateo | Angélica | 422 | 4607/4683 | acantarero(at)cbm.csic.es | Técnico Sup. Actividades Téc. y Profes. GP3 |
| Errahmani Talby | Najlae | 422 | 4683 | Estudiante TFG | |
| García Arias | Juan Manuel | 422 | 4683 | jm.garcia(at)cbm.csic.es | Titulado Sup. Actividades Tecn. y Prof.GP1 |
| Morata Pérez | Ginés | 422 | 4696 | gmorata(at)cbm.csic.es | Doctor Vinculado "Ad Honorem" |
| Ruiz Losada | Mireya | 422 | 4607/4683 | mireya.ruiz(at)cbm.csic.es | M3 |
-Pinal N. Martin, M. Medina, I. and Morata G. “Short-term activation of the Jun-N terminal Kinase
pathway iapoptosis-deficient cells of Drosophila induces tumorigenesis” Nature Comm.
10.1038/s41467-018-04000-6 (2018)
-Bloom-Dahl, D., and N. Azpiazu (2018). The Pax protein Eyegone (Eyg) interacts with the pi-RNA
component Aubergine (Aub) and controls egg chamber development in Drosophila.
Developmental Biology 434(2): 267-277
- Martin, R. and Morata,G. “Regenerative response of different regions of Drosophila imaginal discs” Int. J. Dev. Biol. doi.org/10.1387/ijdb.170326gm (2018)
-Lawrence, PA, Casal, J. de Celis J and Morata, G. "A refutation to Abbasi R, Marcus JM. A new A-P
compartment boundary and organizer in holometabolous insect wings."
Scientific Report 019-42668 (2019)
- Iida, C. Ohsawa, S. Taniguchi, K. Yamamoto, M. Morata, G and Igaki, T. “JNK-mediated Slit-Robo
signaling facilitates epithelial wound repair by extruding dying cells” Scientific Reports
DOI:10.1038/s41598-019-56137-(2019)
-Pinal, N. Calleja, M.and Morata G. “Pro-apoptotic and pro-proliferation functions of the JNK pathway of Drosophila: roles in cell competition, tumourigenesis and regeneration”. Open Biol. 9 (3) : 180256. Doi:10.1098/rsob.180256 (2019)
- Morata, G. and Calleja, M. “Cell competition and tumorigenesis in the imaginal discs of Drosophila”
Seminars in Cancer Biology DOI:10.1016/j.semcancer.2019.06.010 (2020)
- Medina, I. Calleja, M. and Morata G. Tumorigenesis and cell competition in Drosophila in the absence of polyhomeotic function PNAS 118 No. 45 e2110062118 (2021)
- Morata, G. “Cell competition: A historical perspective” Develop. Biol doi.org/10.1016/j.ydbio.2021.02.012 (2021)
- Azpiazu, N. and Morata, G. “Chromatin remodeling and retrotransposons activities during regeneration in Drosophila Develop. Biol doi:10.1016/j.ydbio.2021.11.005 (2021)
- Morata, G. “Cell competition: A historical perspective” Develop. Biol
doi.org/10.1016/j.ydbio.2021.02.012 (2021)
- Morata, G. and Lawrence, P. “An exciting period of Drosophila developmental biology: Of imaginal discs,clones, compartments, parasegments and homeotic genes” Develop. Biol doi.org/10.1016/j.ydbio.2022.01.008(2022)




















