Telomeres in cancer and regeneration
Research summary:
Telomeres in cancer and regeneration.
Telomeres are essential components of our genome, protecting it from deterioration and instability. They consist of repetitive DNA sequences and specialized proteins that are located at the ends of our chromosomes. An important aspect of telomere biology is the potential for telomere fusions, which occur when two chromosomes become physically joined at their telomeres. This can trigger a series of genomic rearrangements and oncogenic alterations, ultimately leading to the development of malignant tumors and resistance to chemotherapy. Despite their relevance in tumor evolution, our understanding of the patterns and consequences of telomere fusions in human cancer is still limited. Our group is currently focused on understanding the role of telomeric fusions in cancer and regeneration.
Telomere fusions in cancer. In recent years, we have used whole-genome sequencing data to study the rates and spectrum of somatic telomere fusions (TFs) in over 30 different types of cancer. Our findings indicate that TFs are widespread in human tumors, with rates that vary greatly between and within different types of cancer. In addition to end-to-end fusions, we have identified unique patterns of TFs that are linked to the activity of the alternative lengthening of telomeres (ALT) pathway. One particularly exciting discovery has been the detection of TFs in the blood of cancer patients, offering a potential method for early cancer detection with high specificity and sensitivity. This is especially promising for cancer types such as pancreatic cancer and brain tumors, which currently have limited options for early detection. Through biochemical, molecular, CRISPR/Cas, and genomic (NGS) techniques, we will continue to investigate the occurrence and function of telomere fusions in various human and mouse tumors. Understanding the role of TFs in cancer will be crucial in developing targeted therapies and improving patient outcomes.
Telomere fusions (TF) in cardiomyocyte maturation and heart regeneration. Maturation in many mammalian cells, including cardiomyocytes, is accompanied by an increase in the number of chromosomes, or polyploidization. This increase in chromosome sets may contribute to the limited ability of mature cardiomyocytes to divide after a damage, as polyploid cells are often unable to undergo successful cell division. While the importance of cardiomyocyte polyploidy is well-recognized, the mechanism behind it is not understood. In recent years using a high-content imaging approach, we have discovered that approximately half of postnatal mouse cardiomyocytes that enter mitosis have discernible chromosome bridges. Furthermore, we observed that the ploidy of cardiomyocytes is tracked by an increasing number of fusions between chromosome ends, providing evidence of a direct link between TFs and cardiomyocyte polyploidization. We also found that TFs are a conserved phenomenon, present in the hearts of mice, zebrafish, pigs, and humans. Importantly, zebrafish hearts, which are able to regenerate and have mostly diploid cardiomyocytes, have few TFs. In fact, deleting telomerase to inhibit zebrafish regeneration increased the number of polyploid cardiomyocytes, supporting the idea that cardiomyocyte polyploidization impairs heart regeneration. Together these results identify TFs as a mechanism leading to chromosome bridging and polyploidization and suggest that failed cardiomyocyte cytokinesis originates from a mitotic defect. We are currently investigating whether fine-tuned regulation of telomerase avoids the presence of TFs, increases the number of diploid cardiomyocytes, and extend the cardiomyocyte proliferation and heart regeneration window. Our findings suggest that regulating the presence of TFs may be key to enhancing cardiomyocyte proliferation and heart regeneration.
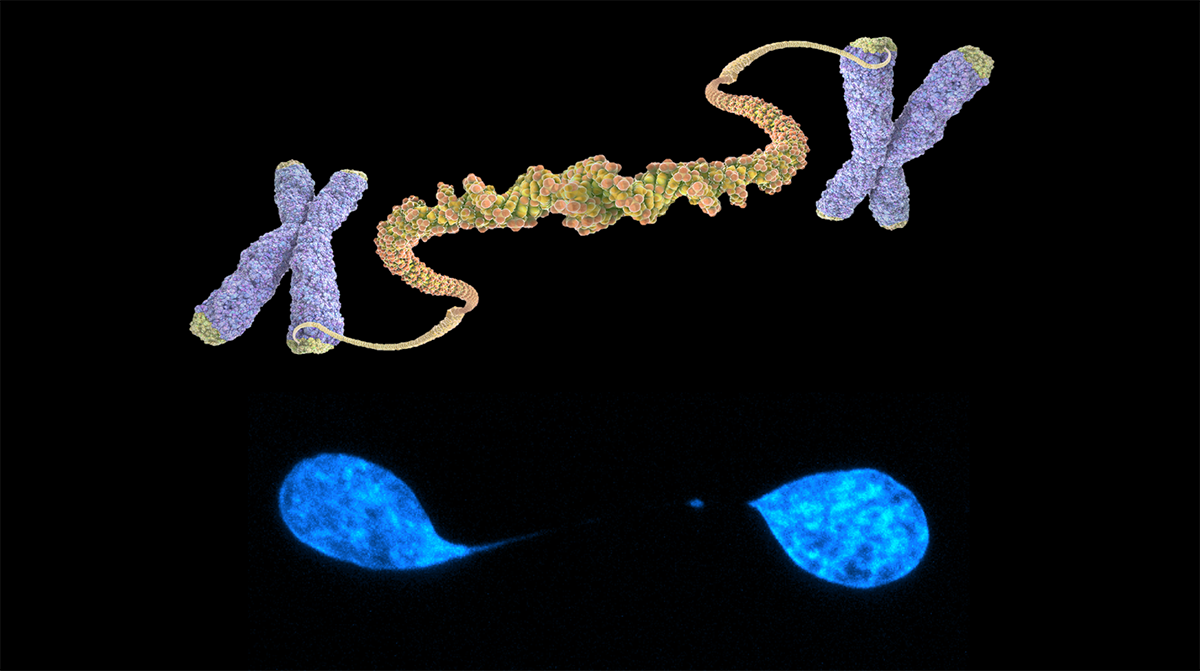
Fig. 1. Telomeric fusion cartoon (up) and a chromatin bridge stained with DAPI in a U2OS cell (bottom).

| Last name | Name | Laboratory | Ext.* | Professional category | |
|---|---|---|---|---|---|
| Flores Hernández | Ignacio | 403/114.1 | 4581 | iflores(at)cbm.csic.es | Investigador Distinguido |
| Morillo Chincoa | María José | 403 | 4608 | maria.morillo(at)cbm.csic.es | M2 |
Relevant Publications:
- Aix E, Gallinat A, Yago-Díez C, Lucas J, Gómez MJ, Benguría A, Freitag P, Cortez-Toledo E, Fernández de Manuel L, García-Cuasimodo L, Sánchez-Iranzo H, Montoya MC, Dopazo A, Sánchez-Cabo F, Mercader N, López JE, Fleischmann BK, Hesse M, Flores I. Telomeres Fuse During Cardiomyocyte Maturation. Circulation. 2023 May 23;147(21):1634-1636.
- González-Estévez C, Flores I. Fasting for stem cell rejuvenation. Aging. 2020 Mar 6;12(5):4048-4049.
- Marín-Aguilar F, Lechuga-Vieco AV, Cañadas-Lozano D, Alcocer-Gómez E, Castejón-Vega B, Lucas J, Garrido C, Peralta-Garcia A, Pérez-Pulido AJ, Varela-López A, Quiles JL, Ryffel B, Flores I, Bullón P, Ruiz-Cabello J, Cordero MD. NLRP3 inflammasome suppression improves longevity and prevents cardiac aging in male mice. Aging Cell. 2020 Jan;19(1):e13050.
- Iglesias M, Felix DA, Gutiérrez-Gutiérrez Ó, De Miguel-Bonet MDM, Sahu S, Fernández-Varas B, Perona R, Aboobaker AA, Flores I*, González-Estévez C*. Downregulation of mTOR Signaling Increases Stem Cell Population Telomere Length during Starvation of Immortal Planarians. Stem Cell Reports. 2019;13(2):405-418. (*Co-corresponding authors)
- Aix E, Gallinat A, Flores I. Telomeres and telomerase in heart regeneration. Differentiation. 2018; 100:26-30.
- Aguado T, Gutiérrez FJ, Aix E, Schneider RP, Giovannazo G, Blasco MA, Flores I. Telomere length defines the cardiomyocyte differentiation potency of mouse induced pluripotent stem cells. Stem Cells. 2017; 35(2):362-37
- Latorre-Pellicer A, Moreno-Loshuertos R, Lechuga-Vieco AV, Sánchez-Cabo F, Torroja C, Acín-Pérez R, Calvo E, Aix E, González-Guerra A, Logan A, Bernad-Miana ML, Romanos E, Cruz R, Carracedo A, Pérez-Martos A, Fernández-Silva P, Ruíz-Cabello J, Murphy M, Flores I, Vázquez J, Enríquez JA. Mismatch between mtDNA and nuclear DNA determines metabolism and healthy ageing. Nature. 2016; 535(7613):561-5.
- Aix E, Gutiérrez-Gutiérrez O and Flores I. Postnatal telomere dysfunction induces postnatal cardiomyocyte cell-cycle arrest through p21 activation. J Cell Biol. 2016; 6; 213(5):571-83. Editor´s choice “In Focus” section PMC4896062.
- Fernández-Alvira JM, Fuster V, Dorado B, Soberón N, Flores I, Gallardo M, Pocock S, Blasco MA and Andrés V. Short telomere load, telomere length and subclinical atherosclerosis in the PESA study. J Am Coll Cardiol. 2016; 67(21):2467-76.
- Bednarek D, González-Rosa JM, Guzmán-Martínez G, Gutiérrez-Gutiérrez Ó, Aguado T, Sánchez-Ferrer C, Marques IJ, Galardi-Castilla M, de Diego I, Gómez MJ, Cortés A, Zapata A, Jiménez-Borreguero LJ, Mercader N, Flores I. Telomerase Is Essential for Zebrafish Heart Regeneration. Cell Rep. 2015; 12(10):1691-703.
- Schneider RP, Garrobo I, Foronda M, Palacios JA, Marión RM, Flores I, Ortega S, Blasco MA. TRF1 is a stem cell marker and is essential for the generation of induced pluripotent stem cells. Nat Commun. 2013; 4:1946.
- Flores I, Blasco MA. Role of telomeres and telomerase in stem cell aging. FEBS Lett. 2010; 584 (17):3826-30. Review.
- Flores I, Blasco MA. A p53-dependent response limits epidermal stem cell functionality and organismal size in mice with short telomeres. PLoS One. 2009; 4(3):e4934.
- Ferrón SR, Marqués-Torrejón MA, Mira H, Flores I, Taylor K, Blasco MA, Fariñas I. Telomere shortening in neural stem cells disrupts neuronal differentiation and neuritogenesis. J Neurosci. 2009; 29(46):14394-407.
- Garcia-Lavandeira M, Quereda V, Flores I, Saez C, Diaz-Rodriguez E, Japon MA, Ryan AK, Blasco MA, Dieguez C, Malumbres M, Alvarez CV. A GRFa2/Prop1/stem (GPS) cell niche in the pituitary. PLoS One. 2009; 4(3):e4815.
- Tomás-Loba A*, Flores I*, Fernández-Marcos PJ, Cayuela ML, Maraver A, Tejera A, Borrás C, Matheu A, Klatt P, Flores JM, Viña J, Serrano M, Blasco MA.Telomerase reverse transcriptase delays aging in cancer-resistant mice. Cell. 2008; 135(4):609-22. (* These authors contributed equally to this work)
- Espada J, Varela I, Flores I, Ugalde AP, Cadiñanos J, Pendás AM, Stewart CL, Tryggvason K, Blasco MA, Freije JM, López-Otín C. Nuclear envelope defects cause stem cell dysfunction in premature-aging mice. J Cell Biol. 2008; 181(1):27-35.
- Flores I, Canela A, Vera E, Tejera A, Cotsarelis G, Blasco MA. The longest telomeres: a general signature of adult stem cell compartments. Genes Dev. 2008; 22(5):654-67.
- Siegl-Cachedenier I, Flores I, Klatt P, Blasco MA.Telomerase reverses epidermal hair follicle stem cell defects and loss of long-term survival associated with critically short telomeres. J Cell Biol. 2007; 179(2):277-90.
- Matheu A, Maraver A, Klatt P, Flores I, Garcia-Cao I, Borras C, Flores JM, Viña J, Blasco MA, Serrano M. Delayed ageing through damage protection by the Arf/p53 pathway. Nature. 2007; 448(7151):375-9.
- Flores I, Evan G, Blasco MA. Genetic analysis of myc and telomerase interactions in vivo. Mol Cell Biol. 2006; 26(16):6130-8.
- Flores I, Benetti R, Blasco MA. Telomerase regulation and stem cell behaviour. Curr Opin Cell Biol. 2006; 18(3):254-60. Review.
- Flores I, Cayuela ML, Blasco MA. Effects of telomerase and telomere length on epidermal stem cell behavior. Science. 2005; 309(5738):1253-6.
- Under review:
- Francesc Muyas, Manuel José Gómez Rodriguez, Isidro Cortes-Ciriano, Ignacio Flores.The ALT pathway generates telomere fusions that can be detected in the blood of cancer patients. bioRxiv 2022.01.25.477771; doi: https://doi.org/10.1101/2022.01.25.477771
- Marielle Breau, Christelle Cayrou, Dmitri Churikov, Charles Fouillade, Sandra Curras-Alonso, Serge Bauwens, Frederic Jourquin, Laura Braud, Frederic Fiore, Rémy Castellao, Emmanuelle Josselin, Carlota SánchezFerrer, Giovanna Giovinazzo, Eric Gilson, Ignacio Flores, Arturo Londono-Vallejo, Serge Adnot, Vincent Géli. Telomerase Prevents Emphysema in Old Mice by Sustaining Subpopulations of Endothelial and AT2 Cells. bioRxiv 2021.01.07.425708; doi: https://doi.org/10.1101/2021.01.07.425708




















