Viral immunology
Research summary:
Our field of interest is aimed at improving the control of chronic and opportunistic infections by the immune system, for which the cellular immune response, as opposed to antibodies alone, plays a leading role, with the long-term goal of contributing to an improved design of new vaccines. Although much is known, vaccines inducing a potent and long-lasting T-cell immunity are still not available. Beyond childhood-oriented vaccines, and due to recent demographic changes and medical advances, the focus of vaccine development is shifting to the new needs of protecting a growing number of adult patients with diverse congenital (primary immunodeficiencies) or iatrogenic levels of immunosuppression (cancer, organ transplantation, autoimmunity) or in specific stages of life (pregnancy, elderly). In order to adapt basic research to these changing needs, we are analyzing basic issues of antiviral cellular immune responses and vaccination in two models of partially immunocompromised mice.
Our first model are mice deficient in the transporter associated with antigen processing (TAP), a crucial player in the MHC class I antigen presentation pathway for recognition by CD8+ T lymphocytes, which are the effector cells responsible for the elimination of infected cells in vivo. These mice are a model for TAP-deficient patients, who suffer from severe recurrent bacterial infections, and have a strong quantitative defect in CD8 cellular immunity, although the remaining CD8+ T lymphocytes are functional. We are currently identifying MHC-I presented viral peptide epitopes, the proteases and the antigen processing pathways generating them in infected cells and trying to elucidate their contribution to the CD8 immune response in these animals.
The second model is represented by mice deficient for the Nras signaling protein, which we have shown to have a qualitative Eomes defect that precludes development of anti-viral memory CD8+ T lymphocytes, and thus prevents vaccination. We are currently evaluating vaccination procedures that could overcome this defect and that might be applied to vaccine design.
The viral infection models we are using are vaccinia virus and mouse cytomegalovirus. Vaccinia virus, a poxvirus, is the vaccine vector behind the first and major success of vaccinology in public health: the eradication of smallpox, officially declared in 1980. Cytomegalovirus is relevant for vaccine design, as it remains latent and continuously presents antigen without pathology, which leads for life to a massive number of functional virus-specific T lymphocytes in the infected organisms, called inflationary memory.
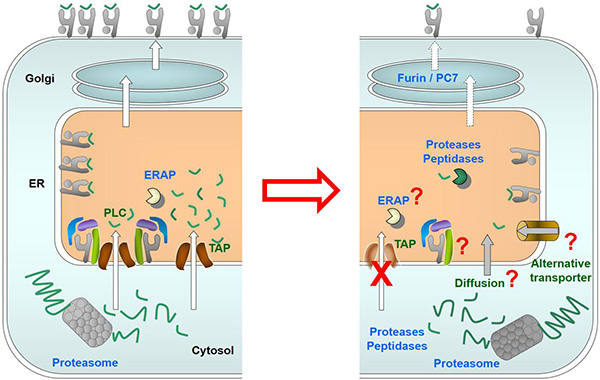
Figure 1. MHC class I viral antigen processing and presentation pathway. The left panel shows the central role played by TAP transporter in this pathway, translocating peptides as well as serving as the hub for the assembly of the peptide loading complex (PLC). The right panel shows the impact of TAP inactivation on MHC class I antigen presentation. The panel also points to potential routes that may mediate the processing of MHC class I antigenic peptides that, although at low levels, are presented by MHC class I in these cells.
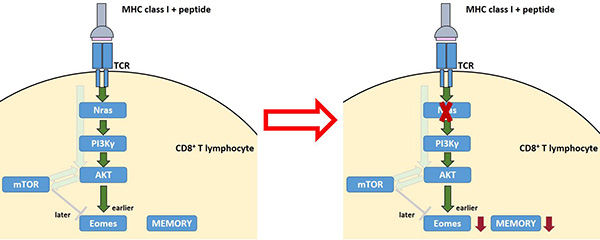
Figure 2. Generation of memory CD8+ cytotoxic T lymphocytes. The left panel shows the T cell receptor (TCR) signaling pathway mediated by Nras that results in the induction of the transcription factor Eomes, which promotes differentiation to memory T lymphocytes. The right panel shows the effect of inactivating Nras in T lymphocytes, which results in a compromised function of Eomes and, consequently a defective memory CD8+ T lymphocyte immune response.

| Last name | Name | Laboratory | Ext.* | Professional category | |
|---|---|---|---|---|---|
| Antón Canto | Luis Carlos | 204 | 4553 | lanton(at)cbm.csic.es | E.Científicos Titulares de Organismos Públicos de Investigación |
| Campos Sánchez | Elena | 204 | 4461 | ecampos(at)cbm.csic.es | Doctor FC2 |
| Fernández Martínez | María | 204 | 4461 | Estudiante | |
| Gutiérrez Oliva | Laura Luna | 204 | 4431 | llgutierrez(at)cbm.csic.es | M3 |
| Ramos Alvarez-Buylla | Manuel | 204 | 4461 | mbuylla(at)cbm.csic.es | E.Científicos Titulares de Organismos Públicos de Investigación |
| Rodríguez Rojas | Cristina | 204 | 4461 | cristina.rrojas(at)cbm.csic.es | M3 |
| Rubio Cachazo | Carolina | 204 | 4461 | carolina.rubio(at)cbm.csic.es | M1 |
| Soto Zaragoza | Andrés | 204 | 4461 | andres.soto(at)cbm.csic.es | Titulado Sup. Actividades Tecn. y Prof.GP1 |
| Val Latorre | Margarita del | 204 | 4460 | mdval(at)cbm.csic.es | E. Investigadores Científicos de Organismos Públicos |
Relevant publications:
- M. Del Val, L.C. Antón, M. Ramos, V. Muñoz-Abad, E. Campos-Sánchez, Endogenous TAP-independent MHC-I antigen presentation: not just the ER lumen, Curr. Op. Immunol. 64 (2020) 9-14.
- J. Navarro, B. Gozalbo-López, A.C. Méndez, F. Dantzer, V. Schreiber, C. Martínez, D.M. Arana, J. Farrés, B. Revilla-Nuin, M.F. Bueno, C. Ampurdanés, M.A. Galindo-Campos, P.A. Knobel, S. Segura-Bayona, J. Martin-Caballero, T.H. Stracker, P. Aparicio, M. Del Val, J. Yélamos, PARP-1/PARP-2 double deficiency in mouse T cells results in faulty immune responses and T lymphomas, Sci. Rep. 7(1) (2017) 41962.
- M. Ramos, Y. Lao, C. Eguiluz, M. Del Val, I. Martínez, Urokinase receptor-deficient mice mount an innate immune response to and clarify respiratory viruses as efficiently as wild-type mice, Virulence 6(7) (2015) 710-715.
- L.C. Antón, J.W. Yewdell, Translating DRiPs: MHC class I immunosurveillance of pathogens and tumors, J. Leuk. Biol. 95(4) (2014) 551-562.
- S. Iborra, M. Ramos, D.M. Arana, S. Lázaro, F. Aguilar, E. Santos, D. López, E. Fernández-Malavé, M. Del Val, N-ras couples antigen receptor signaling to Eomesodermin and to functional CD8+ T cell memory but not to effector differentiation, J. Exp. Med.210(7) (2013) 1463-1479.
- M. Buxadé, G. Lunazzi, J. Minguillón, S. Iborra, R. Berga-Bolaños, M. del Val, J. Aramburu, C. López-Rodríguez, Gene expression induced by Toll-like receptors in macrophages requires the transcription factor NFAT5, J. Exp. Med. 209(2) (2012) 379-393.
- P.S. Satheshkumar, L.C. Anton, P. Sanz, B. Moss, Inhibition of the Ubiquitin-Proteasome System Prevents Vaccinia Virus DNA Replication and Expression of Intermediate and Late Genes, J. Virol. 83(6) (2009) 2469-2479.
- F. Medina, M. Ramos, S. Iborra, P. de León, M. Rodríguez-Castro, M. Del Val, Furin-Processed Antigens Targeted to the Secretory Route Elicit Functional TAP1−/−CD8+ T Lymphocytes In Vivo, J. Immunol. 183(7) (2009) 4639-4647.
- L. Saveanu, O. Carroll, V. Lindo, M. Del Val, D. Lopez, Y. Lepelletier, F. Greer, L. Schomburg, D. Fruci, G. Niedermann, P.M. van Endert, Concerted peptide trimming by human ERAP1 and ERAP2 aminopeptidase complexes in the endoplasmic reticulum, Nature Immunol. 6(7) (2005) 689-697.
- B.C. Gil-Torregrosa, A.R. Castano, M. Del Val, Major histocompatibility complex class I viral antigen processing in the secretory pathway defined by the trans-Golgi network protease furin, J. Exp. Med. 188(6) (1998) 1105-1116.




















