Functional Glycogenomics
Research summary:
The Glycosylation is the most abundant, diverse and dynamic post-translational modification in nature, generating one of the most complex biological molecules found in nature, the glycans. Those are covalent conjugates of an oligosaccharide to certain amino acid residues on the protein backbone, resulting in a plethora of glycoforms potentially exhibiting a wide spectrum of functional and biological proteins for a single gene product. Almost all secreted and membrane proteins are glycosylated and hence almost all plasma and serum proteins are glycoproteins. This co-translational modification widens the functional spectra of proteins at least one magnitude order. Glycan biosynthesis is more significantly affected by disease states than by protein production. Glycomics, therefore holds considerable promise specifically as disease markers. The nonlinear and non-template based biosynthesis of glycans make head to head compare glycomics to proteomics is not technically possible, and complex structural analysis of glycome is necessary in order to get a glycomic profile.
The group has devoted the last five years to assemble, implement and validate a novel technological platform that allows us to analyze the N-glycome from minute amounts of biological samples: sera, plasma or tissues, unique at the UAM campus and second in Spain, and fourth in Europe behind Croacia and Ireland. The group has curated one of the largest collections of clinically well characterized biological samples of American tripanosomiasis biological samples (around 5000), leishmaniasis visceral and Neurocisticercosis from all stages of the diseases, before and after chemotherapeutic treatment.
The glicomic evaluation of individuals (not populations) allows to establish associations to disease progression, therapeutic efficacy or failure and reinfections. The system has been used to analyze samples form three defined infectious disease from which we have clinically defined cohorts (Chagas disease, Leishmaniasis and Neurocysticercosis). From our previous studies on tyiortal sera N-glycome we have moved to study the effector profile of human Immunoglobiulin G derived from its glycosylation profile. By using this novel approach, we have been allowed to identify some molecular markers for efficacy during the treatment with Benznidazole for acute Chagas disease patients, and able to discriminate the latent form active form of neurocysticercosis, previously only possible by using classical image systems like NMR or PET-TAC.
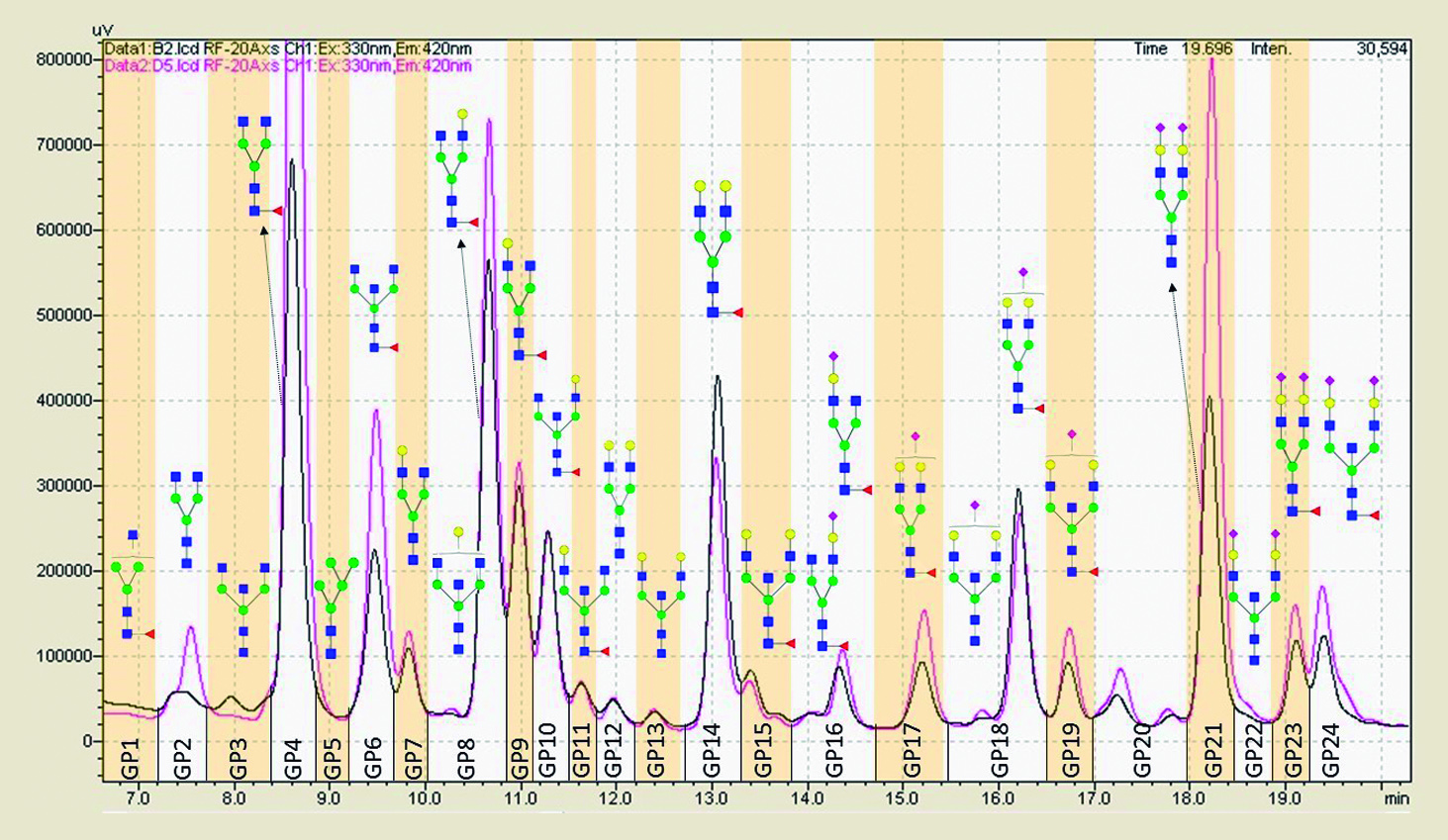
Figure 1: HILIC-UPLC chromatograms stack showing the robustness of glycan resolution from 0.2 uG total IgG isolated from (pink) American trypanosomiasis (Chagas´ disease) patient and (black) control healthy subject

| Last name | Name | Laboratory | Ext.* | Professional category | |
|---|---|---|---|---|---|
| Bonay Miarons | Pedro | 408 | 4509 | pbonay(at)cbm.csic.es | Profesor Titular Universidad, GA |
Relevant publications:
- Pineda M, Corvo L, Callejas-Hernández F, Fresno M, Bonay P. (2020). Trypanosoma cruzi cleaves galectin-3 N-terminal domain to suppress its innate microbicidal activity Clin Exp Immunol. 199(2):216-229. doi: 10.1111/cei.13379.
- Aquilino, C., Gonzalez Rubio, M.L., Seco, E.M., Escudero, L., Corvo, L., Soto, M., Fresno, M., Malpartida, F., and Bonay, P. (2012) Differenti altrypanocidal activity of novel macrolide antibiotics; correlation to genetic lineage. PLoS One 7(7):e40901.
- de la Fuente, H., Pérez-Gala, S., Bonay, P., Cruz-Adalia, A., Cibrian, D., Sánchez-Cuellar, S., Dauden, E., Fresno, M., García-Diez, A., and Sánchez-Madrid, F. (2012). Psoriasis in humans is associated with down-regulation of galectins in dendritic cells. J Pathol. 228(2):193-203.
- Carneiro, M.X., Santos, D.M., Fukutani, K.F., Clarencio, J., Miranda, J.C., Brodskyn, C., Barral, A., Barral-Netto, M., Soto, M. and de Oliveira, C.I. (2012) Vaccination with L. Infantumchagasi nucleosomal histones confers protection against New World Cutaneous Leishmaniasis caused by Leishmania braziliensis. PLoS One 7(12), e52296.
- Valadares, D.G., Duarte, M.C., Ramírez, L., Chávez-Fumagalli, M.A., Martins, V.T., Costa, L.E., Lage, P.S., Ribeiro, T-G., Castilho, R.O., Fernandes, A.P., Régis, W.C., Soto, M., Tavares, C.A. and Coelho, E.A. (2012) Prophylactic or therapeutic administration of Agaricusblazei murillis effective in treatment of murine visceral leishmaniasis. Exp. Parasitol., 132, 228-236.
- Valadares, D.G., Duarte, M.C.m Ramírez, L., Chávez-Fumagalli, M.A., Lage, P.S., Martins, V.T., Costa, L.E., Ribeiro, T.G., Régis, W.C., Soto, M., Fernandes, A.P., Tavares, C.A. and Coelho, E.A. (2012) Therapeutic efficacy induced by the oral administration of Agaricusblazei Murill against Leishmania amazonensis. Parasitol. Res. 111, 1807-1816.
Patents:
- Carlos Alonso, Manuel Soto, Laura Ramírez, Jerónimo Carnés, Marta Román. Molecule for treating an inflammatory disorder. Publication number: WO 2012/152792 A1 (15/11/2012).Laboratorios CBF LETI S.L. Unipersonal.
- Carlos Alonso, Laura Ramírez, Manuel Soto. The use of an L3 and/or L5 source as a vaccine or as a diagnostic for a parasitic disease. Publication number: WO 2011/058137 A1 (15/05/2011). Laboratorios CBF LETI S.L. Unipersonal.
- Manuel Soto, Laura Ramírez and Carlos Alonso. "Diagnosis of a parasitic disease such as leishmaniasis using ribosomal protein extracts (RPE)". Publication number: WO 2011/006891 A1( 20/01/2011). Laboratorios CBF LETI S.L. Unipersonal.
Other activities:
- Pedro Bonay Miarons ha sido nombrado representante español en “International Glycoconjugate Organization”.




















