Molecular pathophysiology of fibrosis
Research summary:
Organ fibrosis is a final common outcome for many diseases such as diabetic nephropathy, liver cirrhosis, scleroderma or myocardial sclerosis. It involves the replacement of cellular living tissue by extracellular matrix, with the subsequent functional derangement. Thus, it is crucial to understand the underlying molecular pathways leading to fibrogenesis in human pathology. MicroRNAs are essential post-transcriptional regulators of gene expression for multiple physiological and pathophysiological pathways, including fibrosis. During the past years we have focused on renal fibrosis regarding two questions: a) the role of metabolism in the genesis of renal injury and repair and b) the role of miRNAs involved in specific metabolic pathways critical for tubular epithelial cell function. We aim to prevent, defer or revert fibrosis in the kidney by understanding underlying metabolic changes associated to kidney fibrosis. To this end we use animal models with specific gain-of-function for critical enzymes involved in fatty acid oxidation, such as carnitine palmitoyltransferase 1A (CPT1A). These studies are complemented by cellular models and biochemical approaches directed towards the study of mitochondrial biogenesis and function. We have found that overexpression of the enzyme Cpt1a in kidney tubules promotes enhanced fatty acid oxidation, restores mitochondrial homeostasis and protects from fibrosis. In addition, we have identified specific microRNAs that are important to regulate the genesis of fibrosis by targeting specific metabolic routes.
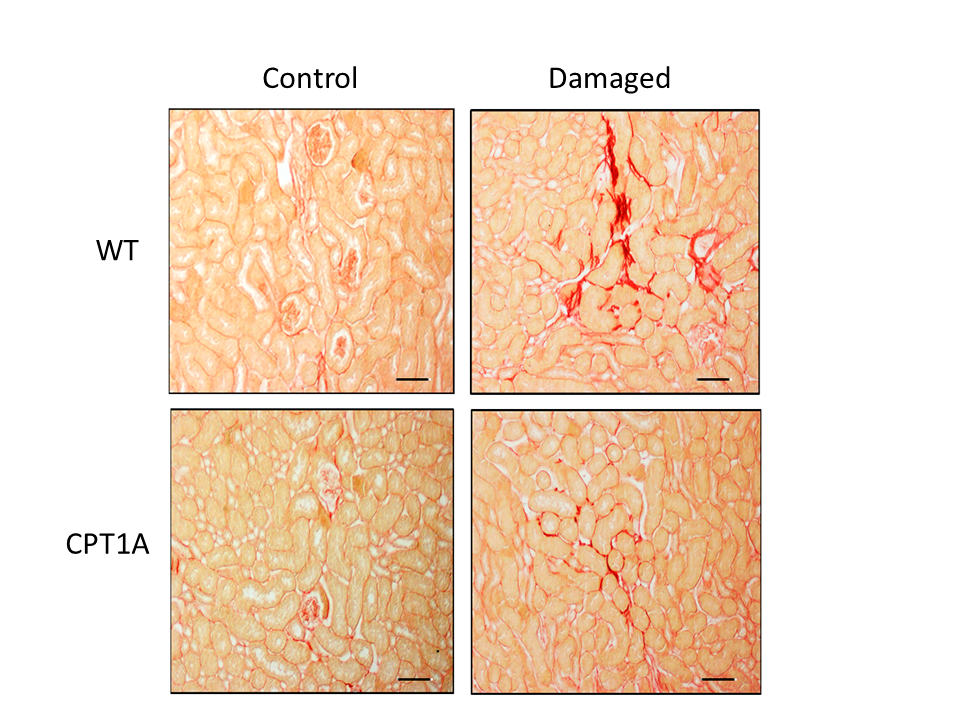
Figure 1: Animal model of kidney fibrosis. Collagen staining with sirius red of kidneys from mice overexpressing Cpt1a in an animal model of experimental fibrosis. Intensity of red color reflects the degree of fibrosis.
* Associate editor of the Redox Biology magazine http://www.journals.elsevier.com/redox-biology/

| Last name | Name | Laboratory | Ext.* | Professional category | |
|---|---|---|---|---|---|
| Jiménez Sánchez | Raquel | 427 | 4740 | rjimenez(at)cbm.csic.es | M1 |
| Lamas Peláez | Santiago | 103 | 4455 | slamas(at)cbm.csic.es | E. Profesores de Investigación de Organismos Públicos de Investigación |
| Romero Albillo | Verónica | 103 | 4505 | vromero(at)cbm.csic.es | M2 |
| Sirera Conca | Belén | 103 | 4456 | bsirera(at)cbm.csic.es | Titulado Sup. Actividades Tecn. y Prof.GP1 |
| Tituaña Fajardo | Jessica Paola | 103 | 4456 | jtituana(at)cbm.csic.es | M2 |
Relevant publications:
- Miguel V, Tituaña J, Herrero JI, Herrero L, Serra D, Cuevas P, Barbas C, Rodríguez-Puyol D, Márquez-Exposito L, Ruiz-Ortega M, Castillo C, Sheng X, Susztak K, Ruiz-Canela M, Salas-Salvadó J, Hu FB, Martínez Gonzalez MA, Ortega S, Ramos R, Lamas S. (2021). Renal tubule Cpt1a overexpression mitigates kidney fibrosis by restoring mitochondrial homeostasis. Journal of Clinical Investigation. 131(5):e140695.
- Miguel V, Ramos R, García-Bermejo L, Rodríguez-Puyol D, Lamas S. (2020). The program of renal fibrogenesis is controlled by microRNAs regulating oxidative metabolism. Redox Biology. 40: 101851.
- Ruiz-Ortega M, Rayego-Mateos S, Lamas S, Ortiz A, Rodrigues-Díez R. (2020). Targeting the progression of chronic kidney disease. Nature Reviews Nephrology. 16, (5):269-288.
- Price NL*, Miguel V*, Ding W, Singh AK, Malik S, Rotllan N, Moshnikova A, Toczek J, Zeiss C, Sadeghi M, Arias Rueda N, Baldán A, Andreev O, Rodriguez-Puyol D, Bahal R, Reshetnyak YK, Suárez Y, Fernández-Hernando C, Lamas S. (2019). Genetic deficiency and pharmacological inhibition of miR-33 enhances renal fatty acid oxidation and attenuates kidney fibrosis. JCI Insight. 4 (22): e131102.
- Fierro-Fernández M, Miguel V, Márquez-Expósito L, Nuevo-Tapioles C, Herrero JI, Blanco-Ruiz E, Tituaña J, Castillo C, Cannata P, Monsalve M, Ruiz-Ortega M, Ramos R, Lamas S. (2020). MiR-9-5p protects from kidney fibrosis by metabolic reprogramming. FASEB Journal. 34(1):410-431.
- Miguel V, Lamas S. (2019) Redox Distress in Organ Fibrosis: The Role of Non-Coding RNAs. Oxidative Stress: Eustress and Distress. Role in Health and Disease Processes. Helmut Sies (Ed.) Elsevier. 37:779-820.
- Espinosa-Díez C, Miguel V, Vallejo S, Sánchez FJ, Sandoval E, Blanco E, Cannata P, Peiró C, Sánchez-Ferrer CF, Lamas S. (2018). Role of glutathione biosynthesis in endothelial dysfunction and fibrosis. Redox Biol. 14:88-99.
- Miguel V, Busnadiego O, Fierro-Fernandez M, Lamas S. (2016). Protective role of miR-9-5p in the fibrogenic transformation of human dermal fibroblasts. Fibrogenesis and Tissue Repair. 9:7.
Doctoral theses:
- Carlos Rey Serra: Impact of the circadian regulation on the metabolic basis of the kidney fibrosis. UAM. Supervisor: Santiago Lamas.
- Alba Sanz Hipólito. (2020). Targeting Durotaxis in Pulmonary Fibrosis and Pancreatic Cancer. UAM. Supervisors: David Lagares Salto and Santiago Lamas Peláez.
- Verónica Miguel Herranz. (2019). The metabolic basis of renal fibrosis: Role of microRNAs and insight from genetic models targeting lipid metabolism. UAM. Supervisor: Santiago Lamas Peláez. International PhD Mention. Awarded with Extraordinary Prize.
- M. Cristina Espinosa Diez. (2015). Role of g-glutamylcysteine ligase in models of vascular and fibrotic damage and the involvement of miR-433 in glutathione synthesis. UAM. Supervisor: Santiago Lamas.
- M. Ángeles Higueras López. (2014). Physiology and Pathophysiology of DLK-1 in the Vascular Endothelium. Universidad Complutense de Madrid. Supervisor: Santiago Lamas.
- Rosa Bretón Romero. (2013). Redox Signaling Responses to Laminar Shear Stress in vascular endothelial cells. UAM. Supervisor. Santiago Lamas
- David Lagares Salto. (2012). Physiopathological and Therapeutic Implications of the TGF-beta / ET-1 axis and Focal Adhesion Kinase in Fibrotic processes. UAM. David Lagares Salto.
Patents:
- European Application "Compounds for prevention and/or treatment of fibrotic diseases" Application No/Patent No 14382239.3-1401
- International application 201031547 “Uso de Dlk1 como inhibidor de angiogénesis”, Dec 2011.
Awards
- Doctor Honoris Causa by the Faculty of medicine of the University of the Republic of Montevideo. 2021.
- Iñigo Álvarez de Toledo nephrology research award. Dr. Verónica Miguel Herranz (2021)
Activities I+D
- Chair in the session “SIMPOSIO. Terapia celular en nefrología”, within the framework of the 51st Virtual Congress of the Spanish Society of Nephrology, held from 15th-18th October 2021.
- Chair in The Training School 3 of ITN TREATMENT. “ The metabolic basis of renal fibrosis”. 1st-3rd July 2019.
- Chair in the EMBO-FEBS Lecture Course. “Molecular Mechanisms of Tissue injury, repair and fibrosis” 23th-31th May 2019.
- Chair in Keystone Symposia on Molecular and Cellular Biology. Role of MicroRNAs in the metabolic basis of renal fibrosis Role of MicroRNAs in the metabolic. 3rd-8th March
- Workshop "Redox Biology as a major drive to the understanding of pathophysiology: Contributions from the CONSOLREDOX network". Salamanca. April 2018




















