Vascular inflammation and autophagy
Research summary:
Inflammation is at the basis of a plethora of diseases with very different aetiological origins. Although this immune reaction plays a paramount role during host survival and tissue repair, inflammation must be tightly regulated to avoid excessive tissue damage and the instigation of inflammatory diseases. For instance, uncontrolled recruitment of immune cells from blood vessels to injured tissues, as well as dysregulated vascular leakage, can lead to the development of numerous pathologies, including highly prevalent cardiovascular, metabolic and infectious conditions as well as tumour progression. Therefore, understanding the mechanisms through which the vasculature becomes dysfunctional during pathological inflammation is an area of extremely forefront research interest.
In recent years, autophagy, a metabolic, cytoplasmic quality control and general homeostatic process, has emerged as a central regulator of immune functions. So far, most of this evidence derives from immune cell-autonomous control of inflammation, with autophagy regulating the survival, differentiation, polarization and inflammatory mediator generation of diverse immune cell subsets. Crucially, our group has recently demonstrated that autophagy processes in endothelial cells, the portal governing the entry of leukocytes and macromolecules into tissues, are essential to limit neutrophil tissue recruitment during acute inflammation and associated tissue damage (Reglero-Real et al. Immunity, 2021). However, the molecular mechanisms and nature of the autophagy processes regulating this phenomenon is still unknown. Hence, the main goal of our emerging laboratory is to decipher the molecular mechanisms through which endothelial cell autophagy pathways shape the inflammatory response with the aim of identifying new therapeutic approaches for the regulation of inflammatory disorders. Collectively, we aim to explore the initiating factors, molecular characteristics and pathophysiological relevance of endothelial autophagy processes during acute and chronic inflammation.
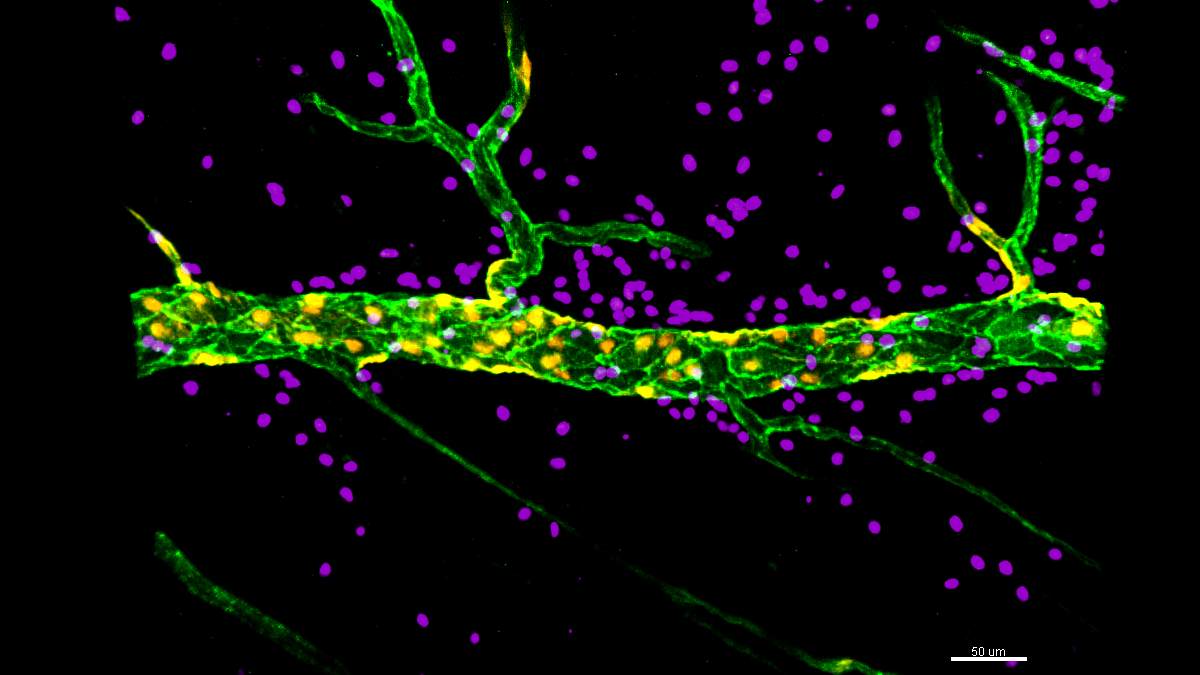
Figure 1. Confocal images of inflamed cremaster muscle postcapillary venules immunostained for endothelial cell junctions (PECAM-1; green) and neutrophils (MRP14; purple).
Movie 1. Confocal intravital microscopy movie of inflamed cremaster muscle postcapillary venules of mice exhibiting wild type (WT; white) or autophagy deficient (magenta; Atg5-/-) endothelial cells. The movie tracks 4 luminal neutrophils (green) migrating through the same Atg5-/- junction and illustrates autophagy deficient junctions have a high capacity to support neutrophil transendothelial migration. Each individual neutrophil migration event is associated with the formation of a transient pore within the PECAM-1 labelled endothelial cell junction.

| Last Name | Name | Laboratory | Ext.* | Professional category | |
|---|---|---|---|---|---|
| Reglero Real | Natalia | 320 | 4652 | nreglero[at]cbm.csic.es | Investigadora Ramón y Cajal |
| García Mateos | Dafne | 320 | 4652 | dafne.garcia[at]cbm.csic.es | Técnico Superior UAM |
| Armesto | Edurne | 320 | 4652 | Estudiante TFG |
Relevant publications:
- Endothelial cell autophagy keeps neutrophil trafficking under control. Reglero-Real N*, Pérez-Gutiérrez L and Sussan Nourshargh. (2021). Autophagy Oct 31:1-3. (* corresponding author) doi: 10.1080/15548627.2021.1987675.
- Autophagy modulates endothelial junctions to restrain neutrophil diapedesis during inflammation. Reglero-Real N†*, Pérez-Gutiérrez L†, Yoshimura A, Rolas L, Garrido-Mesa J, Barkaway A, Pickworth C, Saleeb RS, Gonzalez-Nuñez M, Austin-Williams SN, Cooper D, Vázquez-Martínez L, Fu T, De Rossi G, Golding M, Benoit-Voisin M, Boulanger CM, Kubota Y, Muller WA, Tooze SA, Nightingale TD, Collinson L, Perretti M, Aksoy E and Nourshargh S*. (2021). Immunity Sep 14;54(9):1989-2004. (†co-first author; *co-corresponding author). doi: 10.1016/j.immuni.2021.07.012.
- Age-related cellular and molecular changes in the local inflammatory milieu cause aberrant neutrophil trafficking in vivo. Barkaway A, Rolas L, Joulia R, Bodkin J, Lenn T, Owen-Woods C, Reglero-Real N, Stein M, Vázquez-Martínez L, Girbl T, Poston RN, Golding M, Saleeb RS, Thiriot A, von Andrian UH, Duchene J, Voisin MB, Bishop CL, Voehringer D, Roers A, Rot A, Lämmermann T, Nourshargh S. (2021) Immunity Jul 13;54(7):1494-1510. doi: 10.1016/j.immuni.2021.04.025.
- Time to Take Time Seriously. Reglero-Real N†*, Rolas L†*, Nourshargh S*. (2019) Leukocyte Trafficking: Immunity Feb 19;50(2); 273-275. (†co-first author; *co-corresponding author) doi: 10.1016/j.immuni.2019.01.013.
- Endothelial cell junctional adhesion molecules: Role and Regulation of Expression in inflammation. Reglero-Real N†, Colom B†, Bodkin JV, Nourshargh S. (2016). Arteriosclerosis, Thrombosis and Vascular Biology Oct;36(10); 2048-2057. (†co-first author) # Cover Image of the 2016 Oct. Issue. doi: 10.1161/ATVBAHA.116.307610.
- Cellular Barriers after Extravasation: Leukocyte Interactions with Polarized Epithelia in the Inflamed Tissue. Reglero-Real N, García-Weber D, Millán J. (2016) Mediators of Inflammation 2016; 7650260. doi: 10.1155/2016/7650260.
- Apicobasal polarity controls lymphocyte adhesion to hepatic epithelial cells. Reglero-Real N, Alvarez-Varela A, Cernuda-Morollón E, Feito J, Marcos-Ramiro B, Fernández-Martín L, Gómez-Lechón MJ, Muntane J, Sandoval P, Majano PL, Correas I, Alonso MA and Millán J. (2014) Cell Reports Sep 25;8(6); 1879-1893. doi: 10.1016/j.celrep.2014.08.007.
- Endothelial membrane reorganization during leukocyte extravasation. Reglero-Real N, Marcos-Ramiro B, Millan J. (2012). Cellular and Molecular Life Sciences, Sep;69-18; 3079-3099. doi: 10.1007/s00018-012-0987-4.




















