Neuronal repair and molecular therapy in neurodegeneration: spinocerebellar ataxias
Research summary:
Our group is interested in the study of neurodegenerative diseases. Among them, ataxias are characterized by the loss of neurons in the cerebellum and the spinal cord.
In particular, we have focused on Friedreich’s ataxia, which is the most common hereditary ataxia in the European population. Our aims are the understanding of the mechanisms underlying this pathology and the development of novel therapies for its treatment.
Friedreich's ataxia is an autosomal recessive hereditary disorder resulting from a deficiency of frataxin, which is a protein mainly localized to mitochondria. In addition to neurodegeneration, many patients also suffer from musculoskeletal problems, hypertrophic cardiomyopathy and diabetes. Friedreich´s ataxia is a degenerative disease with a very early onset, and it may serve as a very useful model for other degenerative diseases in which mitochondrial dysfunction also plays an important role.
In a broader context, we are also interested in the key role of mitochondrial metabolism for neuronal homeostatic signaling and of its alterations in neurological diseases.
Our principal research lines include:
1.- Molecular physiopathology of Friedreich's ataxia (PI: Javier Díaz-Nido)
In the context of Friedreich's ataxia, we have developed distinct neural cell models to study the molecular mechanisms underlying the neurodegenerative process triggered by the frataxin deficiency.
These studies may facilitate the identification of novel therapeutic targets and biomarkers not only for Friedreich´s ataxia but also for other neurological diseases characterized by a prominent mitochondrial dysfunction. These cell models are also being used to test potential therapeutic strategies, particularly those focused on identifying molecules (drugs or genes) capable of compensating for the functional defects induced by the loss of frataxin, or that are capable of efficiently increasing the expression of frataxin. In particular we have focused on neurotrophic factors and drugs able to stimulate their production and/or signaling.
2.- Integration of cell signaling and mitochondrial metabolism (PI: Alfredo Giménez-Cassina)
Mitochondrial dysfunction and metabolic decline play a fundamental role in the onset and development of Friedreich's ataxia and other neurological disorders. Our previous studies have shown that manipulation of the metabolic program in the brain constitutes a promising strategy to treat neurological disorders including epilepsy and some neurodegenerative processes. So the understanding of the mechanisms that modulate mitochondrial function and energy metabolism in the brain may contribute to gain insight into the molecular underpinnings responsible for the pathogenesis of diseases in which mitochondrial dysfunction is a prominent feature. In collaboration with the Karolinska Institute (Stockholm, Sweden), we are currently working on identifying and characterizing signaling mechanisms that regulate mitochondrial activity and energy metabolism in neuronal and glial cells, and their impact on neuronal physiology and viability.
3.- Molecular therapy for Friedreich's ataxia (PI: Javier Díaz-Nido)
Las técnicas de transferencia génica constituyen una posibilidad prometedora para la terapia de la ataxia de Friedreich y otras enfermedades neurogenéticas. Las enormes dificultades técnicas para conseguir una eficiente transferencia de los genes “terapéuticos” a las células “diana” exigen realizar un esfuerzo importante en el diseño y desarrollo de nuevas estrategias de terapia génica en distintos modelos experimentales.
Our group has considered a gene therapy approach for Friedreich's ataxia that involves introducing correct copies of the frataxin gene by administering herpes virus vectors carrying the frataxin cDNA or the entire genomic frataxin "locus". We are now attempting to optimize the route of administration and the delivery and distribution of both viral and non-viral vectors in the spinocerebellar system.
Furthermore, we are also investigating the possible application of vectors that may carry other neuroprotective genes, with an emphasis on neurotrophic factors.
Our Research Group also belongs to the the "Instituto de Investigación Sanitaria Hospital Puerta de Hierro Majadahonda IDIPHIM" (Health Research Institute "Puerta de Hierro Majadahonda"). More information at the web page:
http://www.investigacionpuertadehierro.com/index.php/presentacion/
In addition to our research activity, we are strongly committed with the improvement and innovation of learning/teaching in Biomedicine, the development of outreach and communication activities of biomedical research advances, and the bioethical and social implications of biomedical research.
Javier Diaz-Nido is involved in the teaching of different courses at the Bachelor in Biochemistry and the Master in Molecular Biomedicine at UAM. He also participates in the UAM-FCP Joint Institute for Human Rights, Democracy, Culture of Peace and Nonviolence (DEMOS-PAZ). He is currently the acting Director of the Doctoral School at UAM.
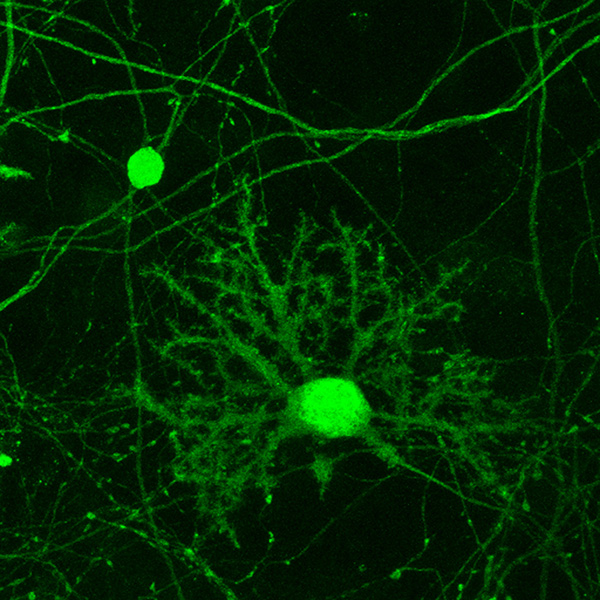
Figure 1.
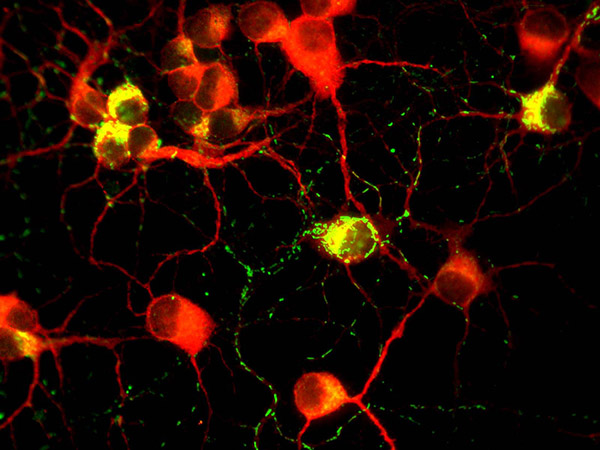
Figure 2.

| Last name | Name | Laboratory | Ext.* | Professional category | |
|---|---|---|---|---|---|
| Díaz Nido | Javier | 207 | 4562 | javier.diaznido(at)cbm.csic.es | Catedrático Universidad, GA |
| Idrovo Gavilanes | Julio César | 207 | 4594 | Estudiante TFM |
Relevant publications:
- Katsu-Jiménez Y, Loria F, Corona JC, Diaz-Nido J. Gene transfer of brain derived neurotrophic factor (BDNF) prevents neurodegeneration triggered by frataxin deficiency. Mol Ther. 2016 Feb 5. doi: 10.1038/mt.2016.32.
- Pérez-Luz S, Gimenez-Cassina A, Fernández-Frías I, Wade-Martins R, Díaz-Nido J. Delivery of the 135 kb human frataxin genomic DNA locus gives rise to different frataxin isoforms. Genomics. 2015 Aug;106(2):76-82. doi: 10.1016/j.ygeno.2015.05.006.
- Loría F, Díaz-Nido J. Frataxin knockdown in human astrocytes triggers cell death and the release of factors that cause neuronal toxicity. Neurobiol Dis. 2015 Apr;76:1-12. doi: 10.1016/j.nbd.2014.12.017.
- Palomo GM, Cerrato T, Gargini R, Diaz-Nido J. Silencing of frataxin gene expression triggers p53-dependent apoptosis in human neuron-like cells. Hum Mol Genet. 2011 Jul 15;20(14):2807-22. doi: 10.1093/hmg/ddr187.
- Gimenez-Cassina A, Wade-Martins R, Gomez-Sebastian S, Corona JC, Lim F, Diaz-Nido J. Infectious delivery and long-term persistence of transgene expression in the brain by a 135-kb iBAC-FXN genomic DNA expression vector. Gene Ther. 2011 Oct;18(10):1015-9. doi: 10.1038/gt.2011.45.
- Gimenez-Cassina A, Lim F, Cerrato T, Palomo GM, Diaz-Nido J. Mitochondrial hexokinase II promotes neuronal survival and acts downstream of glycogen synthase kinase-3. J Biol Chem. 2009 Jan 30;284(5):3001-11. doi: 10.1074/jbc.M808698200.




















