Functional genomics
Research summary:
The central research aim in our group is to understand how genome activity is regulated during early stages of mammalian development and how regulatory and genome structure variations can contributes to human disease. For this, we search for and identify distal acting cis-regulatory sequences, and study how they act on their target genes and how these are organized in the 3D structure of the chromatin and in gene regulatory networks underlying a specific biological state. Methodologically, we use a combination of bioinformatics and data analysis tools, structural genomics, genome-wide analysis, gene-editing using the CRISPR/Cas9 system, and functional tests using transgenic animal models and stem cells.
We are applying this approach to the study of four basic questions:
- How are lineage decisions taken, starting from the fertilized egg, which result in the specification of the initial cell types of the embryo?
- Which are the regulatory mechanism that drive the transition from pluripotency to lineage commitment along development?
- What is the role of the genome structure in the definition of cellular status, and how does it relate to the regulation of gene expression?
- What is the contribution of regulatory variation to common human diseases?
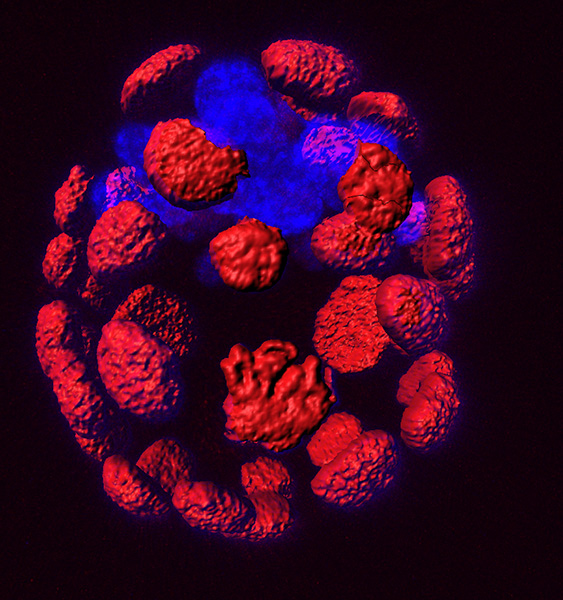
Figure 1. Mouse blastocyst showing expression of CDX2 in red by antibody staining of the outer cells, the trophectoderm, that will produce extraembryonic structures of the embryo such as the placenta. In blue, as shown by nuclear staining with DAPI, is the Inner Cell Mass from where the embryos proper will form.

| Last name | Name | Laboratory | Ext.* | Professional category | |
|---|---|---|---|---|---|
| Arroyo Jiménez | Aurora | 407 | 4474 | aurora.arroyo(at)cbm.csic.es | Titulado Sup. Actividades Tecn. y Prof.GP1 |
| Maidana López | Andrea | 407 | 4474 | amaidana(at)cbm.csic.es | M1 |
| Manzanares Fourcade | Miguel | 407 | 4475 | mmanzanares(at)cbm.csic.es | E. Investigadores Científicos de Organismos Públicos |
| Martín Batista | Elva | 407 | 4474 | embatista(at)cbm.csic.es | Titulado Sup. Actividades Tecn. y Prof.GP1 |
| Portela Martínez | Marta | 407 | 4474 | mportela(at)cbm.csic.es | M2 |
| Tiana Cerrolaza | María | 407 | 4474 | mtiana(at)cbm.csic.es | Investigador |
Relevant publications:
- Menchero S, Rollan I, Lopez-Izquierdo A, Andreu MJ, Sainz de Aja J, Kang M, Adan J, Benedito R, Rayon T, Hadjantonakis AK, Manzanares M (2019). Transitions in cell potency during early mouse development are driven by Notch. eLife 8, e42930.
- Sainz de Aja J, Menchero S, Rollan I, Barral A, Tiana M, Jawaid W, Cossio I, Alvarez A, Carreño-Tarragona G, Badia-Careaga C, Nichols J, Göttgens B, Isern J, Manzanares M (2019). The pluripotency factor NANOG controls primitive hematopoiesis and directly regulates Tal1. EMBO J 38, e99122.
- Gomez-Velazquez M, Badia-Careaga C, Lechuga-Vieco AV, Nieto-Arellano R, Tena JJ, Rollan I, Alvarez A, Torroja C, Fernandez Caceres E, Roy A, Galjart N, Delgado-Olguin P, Sanchez-Cabo F, Enriquez JA, Gomez-Skarmeta JL, Manzanares M (2017). CTCF counter-regulates cardiomyocyte development and maturation programs in the embryonic heart. PLoS Genet 13, e1006985.
- Bogdanovic O, Smits AH, de la Calle Mustienes E, Tena JJ, Ford E, Williams R, Senanayake U, Schultz MD, Hontelez S, van Kruijsbergen I, Rayon T, Gnerlich F, Carell T, Veenstra GJC, Manzanares M, Sauka-Spengler T, Ecker JR, Vermeulen M, Gómez-Skarmeta JL, Lister R (2016). Active DNA demethylation at enhancers during the vertebrate phylotypic period. Nat Genet 48, 417-26.
- Aguirre LA, Alonso ME, Badia-Careaga C, Rollan I, Arias C, Fernandez-Miñan A, López-Jiménez E, Aránega A, Gomez-Skarmeta JL, Franco D, Manzanares M (2015). The 4q25 atrial fibrillation risk locus contains long-range regulatory elements acting on PITX2c and ENPEP. BMC Biol 13, 26.
- Smemo S, Tena JJ, Kim KH, Gamazon ER, Sakabe NJ, Gómez-Marín C, Aneas I, Credidio FL, Sobreira DR, Wasserman NF, Lee JH, Puviindran V, Tam D, Shen M, Son JE, Vakili NA, Sung HK, Naranjo S, Acemel RD, Manzanares M, Nagy A, Cox NJ, Hui CC, Gomez-Skarmeta JL, Nobrega MA (2014). Obesity-associated variants within FTO form long-range functional connections with IRX3. Nature 507, 371-5.
- Rayon T, Menchero S, Nieto A, Xenopoulos P, Crespo M, Cockburn K, Cañon S, Sasaki H, Hadjantonakis AK, de la Pompa JL, Rossant J, Manzanares M (2014). Notch and Hippo converge on Cdx2 to specify the trophectoderm lineage in the mouse blastocyst. Dev Cell 30, 410-422.
- Abad M, Mosteiro L, Pantoja C, Cañamero M, Rayón T, Ors I, Graña O, Megías D, Domínguez O, Martínez D, Manzanares M, Ortega S, Serrano M (2013). Reprogramming in vivo produces teratomas and iPSCs with totipotency features. Nature 502, 340-5.
- Spruce S#, Pernaute B#, Di-Gregorio A, Cobb B, Merkenschlager M, Manzanares M*, Rodriguez TA* (2010). An early developmental role for miRNAs in the maintenance of extra-embryonic stem cells in the mouse embryo. Dev Cell 19, 207-219 (*co-corresponding authors).
- Fernandez-Tresguerres B, Cañon S, Pernaute B, Rayon T, Crespo M, Torroja C, Manzanares M (2010). Evolution of the mammalian embryonic pluripotency gene regulatory network. PNAS 107, 19955-19960.




















