Metabolism and B cell function
Research summary:
In our organism, infection leads to the orchestration of a finetuned immune response. This response relies on the activation of several immune cells; among them, activation of B lymphocytes is crucial, giving place to the production of specific high-affinity antibody-producing cells (plasma cells, PC) and memory B cells (MBC). Activated B cells proliferate massively during the immune response, originating a macroscopic structure within the follicles of secondary lymphoid organs known as Germinal Centers (GC). In GCs, B cells interact with different immune cells in a highly coordinated way which drives their differentiation into PC and MBC.
Deciphering molecular mechanisms controlling GC initiation has attracted the attention of immunologists in the last decade, aiming to understand the stepwise immune response and improve vaccination strategies. In this regard, every time is more evident that GC initiation entails critical metabolic changes in the immune cells. Thus, characterisation of the metabolic profile of each immune cell and understanding how metabolism instructs intrinsic cell activation, proliferation, and differentiation in the GC has become an essential field of study. However, GC is a double-edged sword: while a deficient GC initiation can lead to immunodeficiencies, an overactivation of GC can lead to GC-derived lymphomas or autoimmune diseases. Therefore, in addition to GC initiation, the processes leading to the GC reaction and termination of the cellular expansion are also of high scientific and clinical interest. However, the mechanisms for this GC shutdown have been disregarded until now, considering them as just a passive process.
Our laboratory has generated ground-breaking results showing that GC shutdown is actively triggered by a precise shift of the metabolic profile of GC B cells. Moreover, our data suggest that the metabolic activity of B lymphocytes not only impacts B cells themselves but instructs the metabolic rewiring and fate differentiation of neighbouring T lymphocytes. We hypothesise that massive proliferation at the GC must lead to changes in the substrate availability at the GC microenvironment. These changes may trigger a precise metabolic rewiring in B cells, establishing what we have named as a ¨shutdown metabolic profile¨. This metabolic profile leads to the production of particular signalling molecules, which will both shape B cell function intrinsically and will signal on neighbouring immune cells, driving, all together, the GC shutdown.
Our group aims to characterise the ¨shutdown metabolic profile¨, how it is triggered, and identify all the molecular entities that mediate it. The obtained information will be evaluated in terms of its therapeutic potential in B cell lymphomas and leukaemias, autoimmune diseases, and to improve, in the future, vaccination strategies.
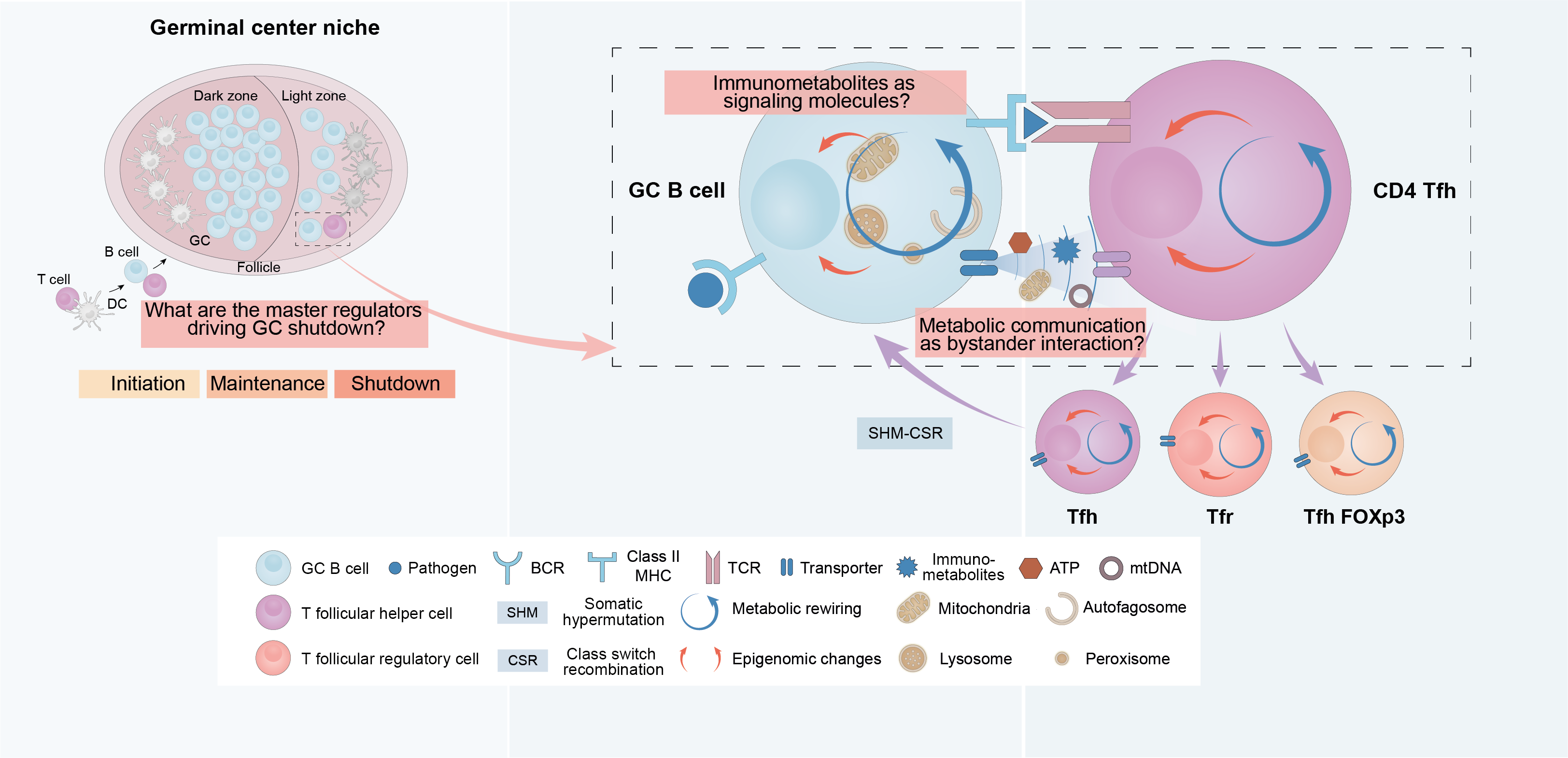
Figure 1: Main questions driving the work at the ¨Metabolism and B cell function laboratory¨

| Last name | Name | Laboratory | Ext.* | Professional category | |
|---|---|---|---|---|---|
| Iborra Pernichi | Marta | 120 | 4721 | marta.iborra(at)cbm.csic.es | Titulado Sup. Actividades Tecn. y Prof.GP1 |
| Martínez Martín | Nuria | 120 | 4517 | nmartinez(at)cbm.csic.es | E.Científicos Titulares de Organismos Públicos de Investigación |
| Quintana Quintana | Carlos | 120 | 4615 | Estudiante | |
| Rodríguez Bovolenta | Elena Giulia | 120 | 4721 | erbovolenta(at)cbm.csic.es | M3 |
| Ruiz García | Jonathan | 120 | 4517 | jruiz(at)cbm.csic.es | M3 Predoc.formación |
| Seco Estrada | Belén | 120 | 4721 | belen.seco(at)cbm.csic.es | M3 Predoc.formación |
| Velasco de la Esperanza | María | 120 | 4721 | mvelasco(at)cbm.csic.es | M1 |
Relevant publications:
- Ortega-Molina A, Deleyto-Seldas N, Carreras J, Sanz A, Lebrero-Fernández C, Menéndez C, Vandenberg A, Fernández-Ruiz B, Marín-Arraiza L, de la Calle Arregui C, Plata-Gómez AB, Caleiras E, de Martino A, Martínez-Martín N, Troulé K, Piñeiro-Yáñez E, Nakamura N, Araf S, Victora GD, Okosun J, Fitzgibbon J, and Efeyan A. (2019). Oncogenic Rag GTPase signalling enhances B cell activation and drives follicular lymphoma sensitive to pharmacological inhibition of mTOR. Nature Metabolism. 1(8);775-789. doi: 10.1038/s42255-019-0098-8.
- Ana Martínez Riaño; Elena Rodriguez Bovolenta; Pilar Mendoza; Clara Oeste; María Jesús Martín Bermejo; Paola Bovolenta; Martin Turner; Nuria Martínez-Martín1; Balbino Alarcón. Antigen phagocytosis by B cells required for a potent humoral response. EMBO Reports. e46016, 2018. (1 co-corresponding author).
- Carlson Tsui; Paula Maldonado; Beatriz Montaner; Borrroto, A; Balbino Alarcón; Andreas Bruckbauer; Nuria Martínez-Martín; Facundo Batista. Dynamic reorganisation of intermediate filaments coordinates early B-cell activation. Life Science Alliance. 1 - 5, 2018.
- Nuria Martínez Martín*; Carlson Tsui*; Mauro Gaya; Paula Maldonado; Mariam Llorian; Nathalie Legrave; Merja Rossi; James I MacRae; Angus J Cameron; Peter J Parker; Michael Leitges; Andreas Bruckbauer; Facundo Batista. PKC? dictates B-cell fate by regulating mitochondrial remodelling, metabolic reprogramming and heme biosynthesis. Immunity. 48, pp. 1144 - 1159. 2018. (* equally contributed).
- Nuria Martínez Martín*1; Pilar Mendoza*; Pablo Hernansanz Agustin; Pilar Delgado; Manuel Diaz Muñoz; Clara Oeste; Isabel Fernández Pisonero; Ester Castellano; Antonio Martinez Ruiz; Eugenio Santos; Tomohiro Kurosaki; Xose Bustelo; Balbino Alarcón. RRas2 is required for germinal center formation to aid B cells during energetically demanding processes. Science Signaling. 11 - 532, 2018. (* equally contributed; 1 co-corresponding author).
- Nuria Martínez-Martín*1; Paula Maldonado*; Francesca Gasparrini; Bruno Frederico; Carlson Tsui; Mariane Burbage; Shweta Aggarwal; Mauro Gaya; Beatriz Montaner; Harold B.J Jefferies; Selina Keppler; Usha Nair; Yan Zhao; Marie-Charlotte Domart; Lucy Collinson; Andreas Bruckbauer; Sharon Tooze; Facundo Batista. 2017. A switch from canonical to non-canonical autophagy shapes B cell responses. Science. Vol 355 – 6325. Pp 641-647. (* equally contributed; 1 co-corresponding author).
Funding:
- Spanish Ministry of Economy and Business (MINECO) – (Programa Retos de la sociedad, 2018-2021)
- Comunidad de Madrid, Programa de Garantía Juvenil 2019 – Ayudante de Investigación




















