Transcriptional control of sexual differentiation of the nervous system
Research summary:
Identifying and understanding the genetic factors linked to neuropsychiatric disorders is a fundamental goal in neuroscience. The nervous system of male and females is sexually different at the molecular and structural levels. These differences lead to sex biases in the age of onset, prevalence, symptomatology and treatment for nearly every neuropsychiatric disorder. The general aim of our laboratory is to decipher the genetic and molecular mechanisms that control sex dependent configurations of the nervous system. Most sex differences are caused by sex hormones, however there is robust evidence that points to genetic factors also contributing to sexual dimorphisms.
The ancient family of DMRT genes encode conserved transcription factors involved in the regulation of sexual features in every animal species so far studied. However, the role of Dmrts in the vertebrate nervous system has not been profusely investigated. We are approaching this question using genetic and genomic approaches in mice. We propose that DMRTs control sex-specific neuron identities and numbers, connectivity and constriction of sexual transcriptional differences in cooperation with sex-linked factors and sex-specific splicing mechanisms. We are also interested in conserved DMRT functions in humans and how specific DMRTs genetic variants could contribute to the onset and sexual bias of psychiatric disorders. Our work will unravel novel principles of brain sexual differentiation and will generate mouse models to better understand genetic mechanisms that either afford protection or generate vulnerability in the etiology and sexual bias of mental disorders.
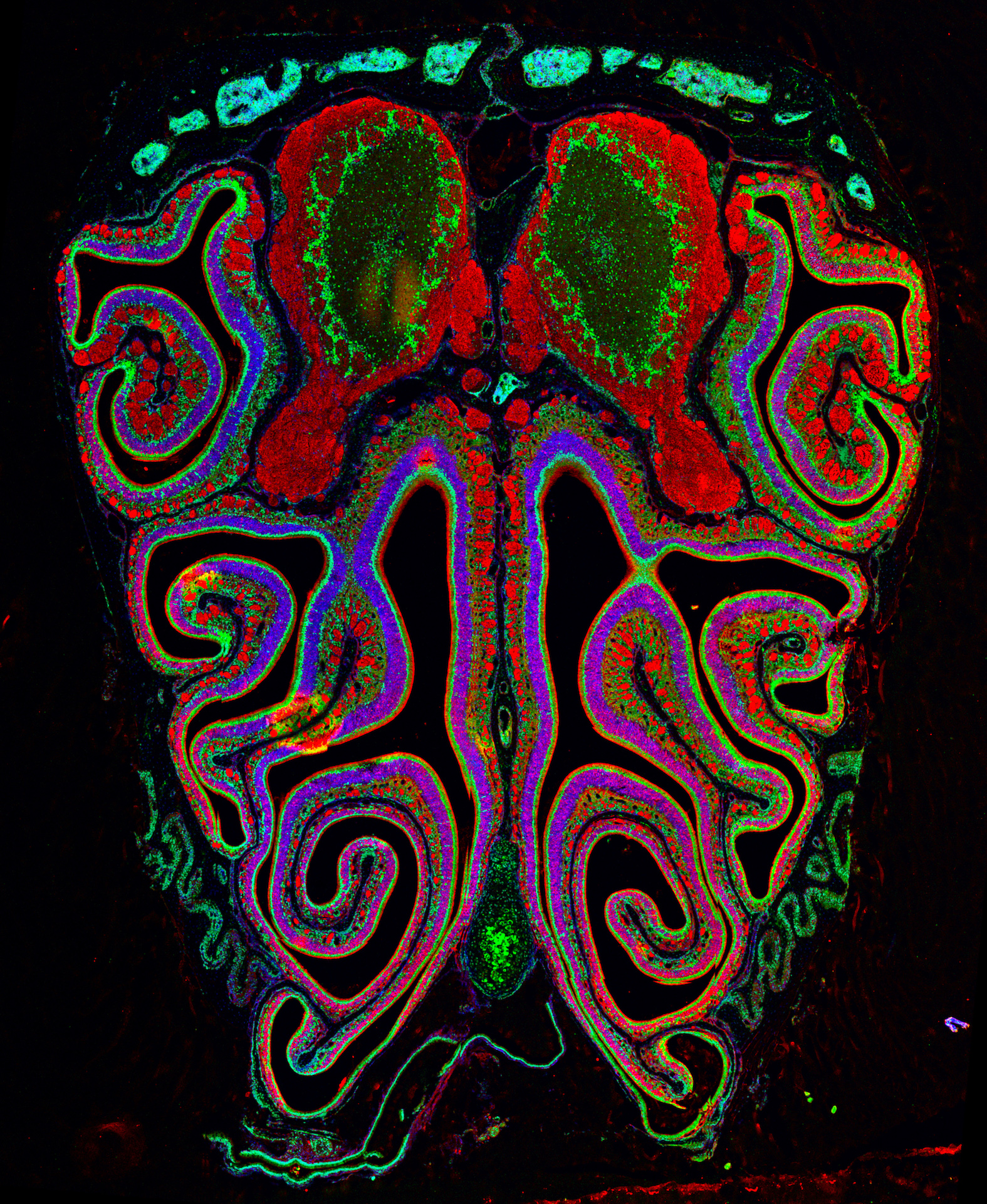
Figure 1. The gates of rodent behaviors (credit: Rafael Casado-Navarro)

| Last name | Name | Laboratory | Ext.* | Professional category | |
|---|---|---|---|---|---|
| Arroyo Almeda | Andrea | 425 | 4464 | aarroyo(at)cbm.csic.es | M3 Predoc.formación |
| Bermejo Santos | Ana | 425 | 4464 | abermejo(at)cbm.csic.es | Titulado Sup. Actividades Tecn. y Prof.GP1 |
| Casado Navarro | Rafael | 425 | 4464 | rcasado(at)cbm.csic.es | Titulado Sup. Actividades Tecn. y Prof.GP1 |
| Serrano Sáiz | Esther | 425 | 4732 | eserrano(at)cbm.csic.es | E.Científicos Titulares de Organismos Públicos de Investigación |
| Torrillas de la Cal | Rodrigo | 425 | 4464 | rtorrillas(at)cbm.csic.es | M3 Predoc.formación |
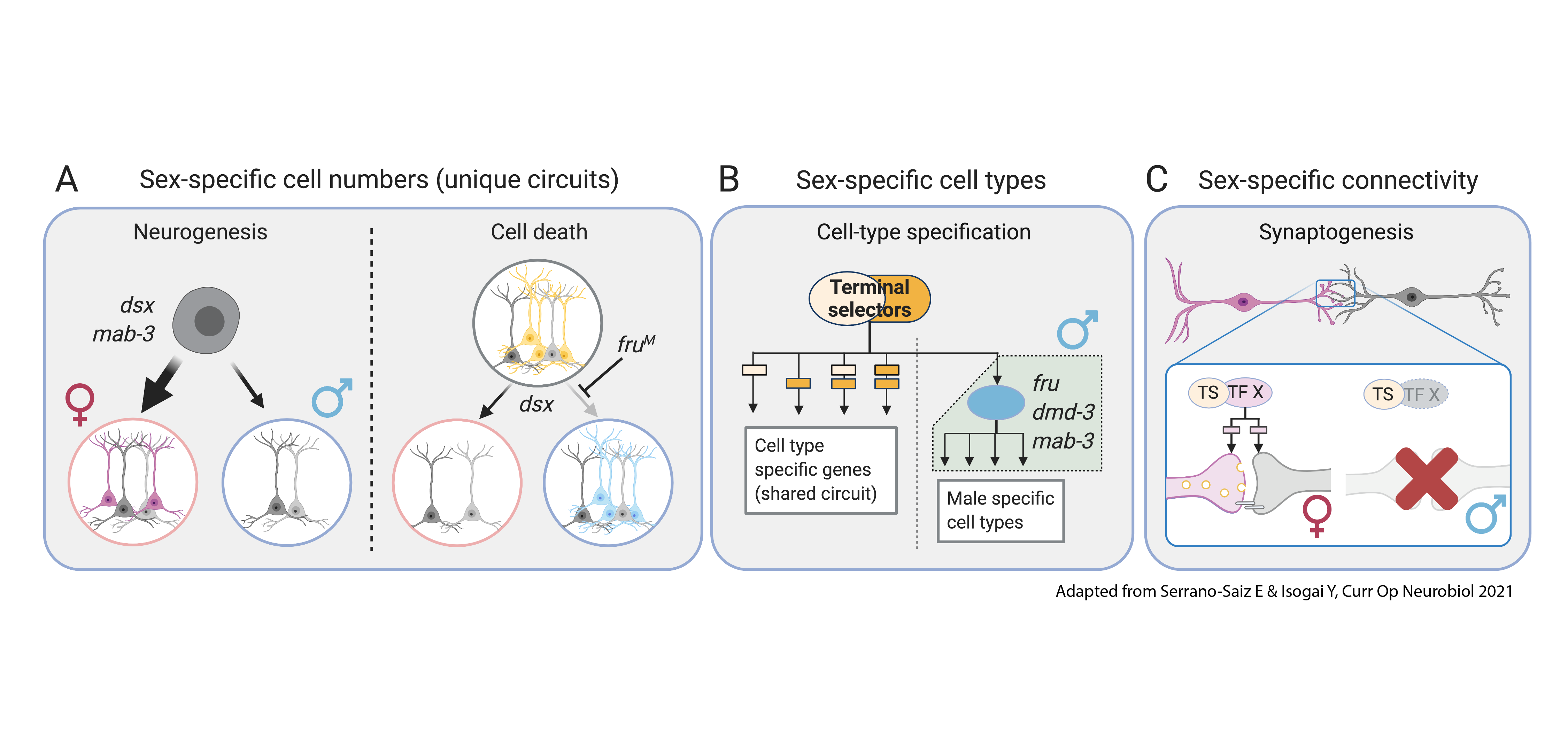
Figure 2. Conserved logic of Dmrt in sexually dimorphic circuits in invertebrate nervous systems (Serrano-Saiz & Isogai, 2021)
Relevant publications:
- Casado-Navarro R and Serrano-Saiz E*. DMRT transcription factors in the control of nervous system sexual differentiation. Front Neuroanat. 2022 Jul 26
- Martín-Fernández F, Bermejo-Santos A, Bragg-Gonzalo L, Briz CG, Serrano-Saiz E, Nieto M. Role of Nrp1 in controlling cortical inter-hemispheric circuits. eLife, 2022 Mar 1;11:e69776.
- Serrano-Saiz E and Isogai Y. Single-cell molecular and developmental perspectives of sexually dimorphic circuits underlying innate behaviors. Curr Opinion in Neurobiol. 2021. Jun;68:159-166
- Pereira L, Aeschimann F, Wang C, Lawson H, Serrano-Saiz E, Portman D, Grosshans H, Hobert O. Timing mechanism of sexually dimorphic nervous system differentiation. eLife, 2019 Jan 1;8.
- Serrano-Saiz E*, Leyva-Diaz E, De la Cruz E, Hobert O*. BRN3A-type POU homeobox genes maintain the identity of mature postmitotic neurons in nematodes and mice. Current Biology, 2018 Sep 10;28(17):2813-2823
- Serrano-Saiz E*, Oren-Suissa M and Hobert O*. Sexually dimorphic differentiation of a elegans hub neuron is cell-autonomously controlled by a conserved transcription factor. Current Biology, 2017 Jan 23;27(2):199-209. (F1000 recommended)
- Serrano-Saiz E*, Poole R, Felton T, Feifan Zhang, De la Cruz E, Hobert O*. Modular control of glutamatergic neuronal identity in C. elegans by distinct homeodomain proteins. Cell, 2013 Oct 24;155(3):659-73
Funding:
- Fundación Alicia Koplowitz 2022
- ReDevNeural 3.0
- Spanish Ministry of Science and Innovation (MCIN) – (Programa Generación de conocimiento 2021)
Other merits:
- Chapter coordinator CSIC White Book - The mosaic brain. Sex/gender and the neurosciences
- Member of the CBMSO Equality group
- Seminar organizer of Tissue and Organ Homeostasis




















